Klotho and activin A in kidney injury: plasma Klotho is maintained in unilateral obstruction despite no upregulation of Klotho biosynthesis in the contralateral kidney
- PMID: 29187373
- PMCID: PMC6031917
- DOI: 10.1152/ajprenal.00528.2017
Klotho and activin A in kidney injury: plasma Klotho is maintained in unilateral obstruction despite no upregulation of Klotho biosynthesis in the contralateral kidney
Abstract
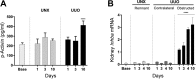
In a new paradigm of etiology related to chronic kidney disease-mineral and bone disorder (CKD-MBD), kidney injury may cause induction of factors in the injured kidney that are released into the circulation and thereby initiate and maintain renal fibrosis and CKD-MBD. Klotho is believed to ameliorate renal fibrosis and CKD-MBD, while activin A might have detrimental effects. The unilateral ureter obstruction (UUO) model is used here to examine this concept by investigating early changes related to renal fibrosis in the obstructed kidney, untouched contralateral kidney, and vasculature which might be affected by secreted factors from the obstructed kidney, and comparing with unilateral nephrectomized controls (UNX). Obstructed kidneys showed early Klotho gene and protein depletion, whereas plasma Klotho increased in both UUO and UNX rats, indicating an altered metabolism of Klotho. Contralateral kidneys had no compensatory upregulation of Klotho and maintained normal expression of the examined fibrosis-related genes, as did remnant UNX kidneys. UUO caused upregulation of transforming growth factor-β and induction of periostin and activin A in obstructed kidneys without changes in the contralateral kidneys. Plasma activin A doubled in UUO rats after 10 days while no changes were seen in UNX rats, suggesting secretion of activin A from the obstructed kidney with potentially systemic effects on CKD-MBD. As such, increased aortic sclerostin was observed in UUO rats compared with UNX and normal controls. The present results are in line with the new paradigm and show very early vascular effects of unilateral kidney fibrosis, supporting the existence of a new kidney-vasculature axis.
Keywords: Klotho; Wnt; activin A; renal fibrosis; unilateral ureter obstruction.
Figures



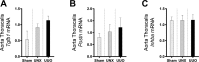
References
-
- Barker SL, Pastor J, Carranza D, Quiñones H, Griffith C, Goetz R, Mohammadi M, Ye J, Zhang J, Hu MC, Kuro-o M, Moe OW, Sidhu SS. The demonstration of αKlotho deficiency in human chronic kidney disease with a novel synthetic antibody. Nephrol Dial Transplant 30: 223–233, 2015. doi: 10.1093/ndt/gfu291. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

