The NADPH organizers NoxO1 and p47phox are both mediators of diabetes-induced vascular dysfunction in mice
- PMID: 29195137
- PMCID: PMC5723277
- DOI: 10.1016/j.redox.2017.11.014
The NADPH organizers NoxO1 and p47phox are both mediators of diabetes-induced vascular dysfunction in mice
Abstract
Aim: NADPH oxidases are important sources of reactive oxygen species (ROS). Several Nox homologues are present together in the vascular system but whether they exhibit crosstalk at the activity level is unknown. To address this, vessel function of knockout mice for the cytosolic Nox organizer proteins p47phox, NoxO1 and a p47phox-NoxO1-double knockout were studied under normal condition and during streptozotocin-induced diabetes.
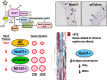
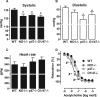
Results: In the mouse aorta, mRNA expression for NoxO1 was predominant in smooth muscle and endothelial cells, whereas p47phox was markedly expressed in adventitial cells comprising leukocytes and tissue resident macrophages. Knockout of either NoxO1 or p47phox resulted in lower basal blood pressure. Deletion of any of the two subunits also prevented diabetes-induced vascular dysfunction. mRNA expression analysis by MACE (Massive Analysis of cDNA ends) identified substantial gene expression differences between the mouse lines and in response to diabetes. Deletion of p47phox induced inflammatory activation with increased markers of myeloid cells and cytokine and chemokine induction. In contrast, deletion of NoxO1 resulted in an attenuated interferon gamma signature and reduced expression of genes related to antigen presentation. This aspect was also reflected by a reduced number of circulating lymphocytes in NoxO1-/- mice.
Innovation and conclusion: ROS production stimulated by NoxO1 and p47phox limit endothelium-dependent relaxation and maintain blood pressure in mice. However, NoxO1 and p47phox cannot substitute each other despite their similar effect on vascular function. Deletion of NoxO1 induced an anti-inflammatory phenotype, whereas p47phox deletion rather elicited a hyper-inflammatory response.
Keywords: NADPH oxidase; Nox1; NoxO1; Reactive oxygen species; Superoxide; p47phox.
Copyright © 2017 The Authors. Published by Elsevier B.V. All rights reserved.
Figures





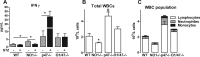
References
-
- Youn J.Y., Gao L., Cai H. The p47phox- and NADPH oxidase organiser 1 (NOXO1)-dependent activation of NADPH oxidase 1 (NOX1) mediates endothelial nitric oxide synthase (eNOS) uncoupling and endothelial dysfunction in a streptozotocin-induced murine model of diabetes. Diabetologia. 2012;55(7):2069–2079. - PMC - PubMed
-
- Paradis P., Ouerd S., Idris-Khodja N., Trindade M., Coelho S.C., Jandeleit-Dahm K.A., Schiffrin E.L. Vol. 34. 2016. OS 21-01 NOX1 OR NOX4 deletion prevents type 1 diabetes-induced endothelial dysfunction; p. e235. (J. Hypertens.). (ISH 2016 Abstract Book)
-
- Lee M.Y., San Martin A., Mehta P.K., Dikalova A.E., Garrido A.M., Datla S.R., Lyons E., Krause K.-H., Banfi B., Lambeth J.D., Lassegue B., Griendling K.K. Mechanisms of vascular smooth muscle NADPH oxidase 1 (Nox1) contribution to injury-induced neointimal formation. Arterioscler. Thromb. Vasc. Biol. 2009;29(4):480–487. - PMC - PubMed
-
- Wind S., Beuerlein K., Armitage M.E., Taye A., Kumar A.H.S., Janowitz D., Neff C., Shah A.M., Wingler K., Schmidt H.H.H.W. Oxidative stress and endothelial dysfunction in aortas of aged spontaneously hypertensive rats by NOX1/2 is reversed by NADPH oxidase inhibition. Hypertension. 2010;56(3):490–497. - PubMed
-
- Matsuno K., Yamada H., Iwata K., Jin D., Katsuyama M., Matsuki M., Takai S., Yamanishi K., Miyazaki M., Matsubara H., Yabe-Nishimura C. Nox1 is involved in angiotensin II-mediated hypertension: a study in Nox1-deficient mice. Circulation. 2005;112(17):2677–2685. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

