Radiopharmaceutical tracers for cardiac imaging
- PMID: 29196910
- PMCID: PMC6133155
- DOI: 10.1007/s12350-017-1131-5
Radiopharmaceutical tracers for cardiac imaging
Erratum in
-
Correction to: Radiopharmaceutical tracers for cardiac imaging.J Nucl Cardiol. 2018 Aug;25(4):1237-1241. doi: 10.1007/s12350-018-1184-0. J Nucl Cardiol. 2018. PMID: 29368086 Free PMC article.
Abstract
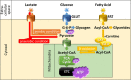
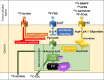
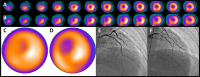
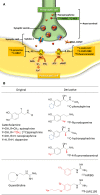
Cardiovascular disease (CVD) is the leading cause of death and disease burden worldwide. Nuclear myocardial perfusion imaging with either single-photon emission computed tomography or positron emission tomography has been used extensively to perform diagnosis, monitor therapies, and predict cardiovascular events. Several radiopharmaceutical tracers have recently been developed to evaluate CVD by targeting myocardial perfusion, metabolism, innervation, and inflammation. This article reviews old and newer used in nuclear cardiac imaging.
Keywords: Cardiovascular disease; positron emission tomography; radiopharmaceutical; single-photon emission computed tomography.
Figures
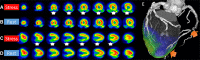
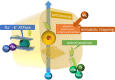
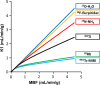
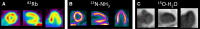
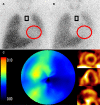
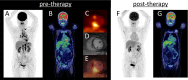
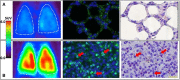

Comment in
-
Radiopharmaceuticals for cardiac imaging: Current status and future trends.J Nucl Cardiol. 2018 Aug;25(4):1242-1246. doi: 10.1007/s12350-018-1194-y. Epub 2018 Feb 7. J Nucl Cardiol. 2018. PMID: 29417412 No abstract available.
References
-
- Bateman TM, Dilsizian V, Beanlands RS, DePuey EG, Heller GV, Wolinsky DA. American Society of Nuclear Cardiology and Society of Nuclear Medicine and Molecular Imaging Joint Position Statement on the Clinical Indications for Myocardial Perfusion PET. J Nucl Med. 2016;57:1654–1656. - PubMed
-
- Masuda A, Takeishi Y. Choosing the appropriate examination for diagnosis of stable ischemic heart disease. Ann Nucl Cardiol. 2016;2:167–173.
-
- Schindler T, Marashdeh W, Solnes L. Application of myocardial blood flow quantification in CAD patients. Ann Nucl Cardiol. 2016;2:84–93.
-
- Rauch B, Helus F, Grunze M, Braunwell E, Mall G, Hasselbach W, Kubler W. Kinetics of 13N-ammonia uptake in myocardial single cells indicating potential limitations in its applicability as a marker of myocardial blood flow. Circulation. 1985;71:387–393. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

