Oxcarbazepine for neuropathic pain
- PMID: 29199767
- PMCID: PMC6486101
- DOI: 10.1002/14651858.CD007963.pub3
Oxcarbazepine for neuropathic pain
Abstract
Background: Several anticonvulsant drugs are used in the management of neuropathic pain. Oxcarbazepine is an anticonvulsant drug closely related to carbamazepine. Oxcarbazepine has been reported to be efficacious in the treatment of neuropathic pain, but evidence from randomised controlled trials (RCTs) is conflicting. Oxcarbazepine is reportedly better tolerated than carbamazepine. This is the first update of a review published in 2013.
Objectives: To assess the benefits and harms of oxcarbazepine for different types of neuropathic pain.
Search methods: On 21 November 2016, we searched the Cochrane Neuromuscular Specialised Register, CENTRAL, MEDLINE and Embase. We searched the Chinese Biomedical Retrieval System (January 1978 to November 2016). We searched the US National Institutes of Health (NIH) databases and the World Health Organization (WHO) International Clinical Trials Registry Platform for ongoing trials in January 2017, and we wrote to the companies who make oxcarbazepine and to pain experts requesting additional information.
Selection criteria: All RCTs and randomised cross-over studies of oxcarbazepine for the treatment of people of any age or sex with any neuropathic pain were eligible. We planned to include trials of oxcarbazepine compared with placebo or any other intervention with a treatment duration of at least six weeks, regardless of administration route and dose.
Data collection and analysis: We used standard methodological procedures expected by Cochrane.
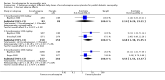
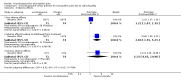
Main results: Five multicentre, randomised, placebo-controlled, double-blind trials with a total of 862 participants were eligible for inclusion in this updated review. Three trials involved participants with painful diabetic peripheral neuropathy (DPN) (n = 634), one included people with neuropathic pain due to radiculopathy (n = 145), and one, which was newly identified at this update, involved participants with peripheral neuropathic pain of mixed origin (polyneuropathy, peripheral nerve injury or postherpetic neuralgia) (n = 83). Some studies did not report all outcomes of interest. For painful DPN, compared to the baseline, the proportion of participants who reported at least a 50% or 30% reduction of pain scores after 16 weeks of treatment in the oxcarbazepine group versus the placebo group were: at least 50% reduction: 34.8% with oxcarbazepine versus 18.2% with placebo (risk ratio (RR) 1.91, 95% confidence interval (CI) 1.08 to 3.39, number of people needed to treat for an additional beneficial outcome (NNTB) 6, 95% CI 3 to 41); and at least 30% reduction: 44.9% with oxcarbazepine versus 28.6% with placebo (RR 1.57, 95% CI 1.01 to 2.44; NNTB 6, 95% CI 3 to 114; n = 146). Both results were based on data from a single trial, since two trials that found little or no benefit did not provide data that could be included in a meta-analysis. Although these trials were well designed, incomplete outcome data and possible unblinding of participants due to obvious adverse effects placed the results at a high risk of bias. There was also serious imprecision and a high risk of publication bias. The radiculopathy trial reported no benefit for the outcome 'at least 50% pain relief' from oxcarbazepine. In mixed neuropathies, 19.3% of people receiving oxcarbazepine versus 4.8% receiving placebo had at least 50% pain relief. These small trials had low event rates and provided, at best, low-quality evidence for any outcome. The proportion of people with 'improved' or 'very much improved' pain was 45.9% with oxcarbazepine versus 30.1% with placebo in DPN (RR 1.46, 95% CI 1.13 to 1.88; n = 493; 2 trials; very-low-quality evidence) and 23.9% with oxcarbazepine versus 14.9% with placebo in radiculopathy (RR 1.61, 95% CI 0.81 to 3.20; n = 145).We found no trials in other types of neuropathic pain such as trigeminal neuralgia.Trial reports stated that most adverse effects were mild to moderate in severity. Based on moderate-quality evidence from the three DPN trials, serious adverse effects occurred in 8.3% with oxcarbazepine and 2.5% with placebo (RR 3.65, 95% CI 1.45 to 9.20; n = 634; moderate-quality evidence). The number needed to treat for an additional harmful (serious adverse effect) outcome (NNTH) was 17 (95% CI 11 to 42). The RR for serious adverse effects in the radiculopathy trial was 3.13 (95% CI 0.65 to 14.98, n = 145). The fifth trial did not provide data.More people withdrew because of adverse effects with oxcarbazepine than with placebo (DPN: 25.6% with oxcarbazepine versus 6.8% with placebo; RR 3.83, 95% CI 2.29 to 6.40; radiculopathy: 42.3% with oxcarbazepine versus 14.9% with placebo; RR 2.84, 95% CI 1.55 to 5.23; mixed neuropathic pain: 13.5% with oxcarbazepine versus 1.2% with placebo; RR 11.51, 95% CI 1.54 to 86.15).
Authors' conclusions: This review found little evidence to support the effectiveness of oxcarbazepine in painful diabetic neuropathy, neuropathic pain from radiculopathy and a mixture of neuropathies. Some very-low-quality evidence suggests efficacy but small trials, low event rates, heterogeneity in some measures and a high risk of publication bias means that we have very low confidence in the measures of effect. Adverse effects, serious adverse effects and adverse effects leading to discontinuation are probably more common with oxcarbazepine than placebo; however, the numbers of participants and event rates are low. More well-designed, multicentre RCTs investigating oxcarbazepine for various types of neuropathic pain are needed, and selective publication of studies or data should be avoided.
Conflict of interest statement
MZ: none known.
NC: none known.
LH: none known.
MY: none known.
CZ: none known.
FW: none known.
Figures
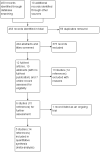













Update of
-
Oxcarbazepine for neuropathic pain.Cochrane Database Syst Rev. 2013 Mar 28;(3):CD007963. doi: 10.1002/14651858.CD007963.pub2. Cochrane Database Syst Rev. 2013. Update in: Cochrane Database Syst Rev. 2017 Dec 02;12:CD007963. doi: 10.1002/14651858.CD007963.pub3. PMID: 23543558 Updated.
References
References to studies included in this review
Beydoun 2006 {published and unpublished data}
-
- Multicenter, double‐blind, randomized placebo‐control, parallel group study to evaluate the safety and efficacy of three doses of oxcarbazepine in patients with neuropathic pain due to diabetic neuropathy, 2003. www.novctrd.com/CtrdWeb/product.nov?diseaseid=141&productid=330 (accessed 2 October 2015).
-
- Beydoun A, Shaibani A, Hopwood M, Wan Y. Oxcarbazepine in painful diabetic neuropathy: results of a dose‐ranging study. Acta Neurologica Scandinavica 2006;113(6):395‐404. [PUBMED: 16674606] - PubMed
-
- Beydoun A, Wan Y, Hopwood M, Liebel J. Results of a randomized, placebo‐controlled trial suggest oxcarbazepine has a therapeutic effect in the treatment of painful diabetic neuropathy. European Journal of Neurology 2004;11 Suppl 2:101.
Demant 2014 {published and unpublished data}
-
- Oxcarbazepine for the treatment of chronic peripheral neuropathic pain (IMIOXC). https://clinicaltrials.gov/ct2/show/study/NCT01302275?term=NCT01302275&a... 2014 (accessed 2 October 2015).
-
- Demant DT, Lund K, Vollert J, Maier C, Segerdahl M, Finnerup NB, et al. The effect of oxcarbazepine in peripheral neuropathic pain depends on pain phenotype: a randomised, double‐blind, placebo‐controlled phenotype‐stratified study. Pain 2014;155(11):2263‐73. [PUBMED: 25139589] - PubMed
Dogra 2005 {published and unpublished data}
-
- Multicenter, double‐blind, randomized placebo‐control, parallel‐group study to evaluate the safety and efficacy of oxcarbazepine in patients with neuropathic pain due to diabetic neuropathy, 2005. www.novctrd.com/CtrdWeb/product.nov?diseaseid=141&productid=330 (accessed 2 October 2015).
-
- Dogra S, Beydoun S, Hopwood M, Mazzola J, Wan Y. Oxcarbazepine in painful diabetic neuropathy: a randomized, placebo‐controlled study. Journal of Pain 2004;5(3 Suppl 1):S6. - PubMed
-
- Dogra S, Beydoun S, Mazzola J, Hopwood M, Wan Y. Oxcarbazepine in painful diabetic neuropathy: a randomized, placebo‐controlled study. European Journal of Pain 2005;9(5):543‐54. [PUBMED: 16139183] - PubMed
-
- Dogra S, Beydoun S, Mazzola J, Hopwood M, Wan Y, Liebel J. Oxcarbazepine provides pain relief in patients with painful diabetic neuropathy: a randomized, placebo‐controlled study. European Journal of Neurology 2004;11 Suppl 2:92.
Grosskopf 2006 {published and unpublished data}
-
- Multicenter, double‐blind, randomized, placebo‐control, parallel study to evaluate the safety and efficacy of oxcarbazepine in patients with neuropathic pain due to diabetic neuropathy, 2003. www.novctrd.com/CtrdWeb/product.nov?diseaseid=141&productid=330 (accessed 2 October 2015).
-
- Grosskopf J, Mazzola J, Wan Y, Hopwood M. A randomized, placebo‐controlled study of oxcarbazepine in painful diabetic neuropathy. Acta Neurologica Scandinavica 2006;114(3):177‐80. [PUBMED: 16911345] - PubMed
-
- Riviere M‐E, Wan Y, Hopwood M, Liebel J. Oxcarbazepine in the treatment of painful diabetic neuropathy: a randomized, placebo‐controlled study. European Journal of Neurology 2004;11 Suppl 2:101.
Novartis 2004 {published and unpublished data}
-
- Multicenter, double‐blind, randomized, placebo‐control, parallel‐group study to evaluate the safety and efficacy of oxcarbazepine in patients with neuropathic pain due to radiculopathy, 2005. www.novctrd.com/CtrdWeb/product.nov?diseaseid=429&productid=330 (accessed 2 October 2015).
-
- Harden RN, Urniasz KL, Hopwood M, Wan Y. Efficacy and safety of oxcarbazepine in patients with neuropathic pain due to radiculopathy: a double‐blind, randomized, placebo‐controlled study. Neurology 2005;64 Suppl 1:A120‐1.
References to studies excluded from this review
Besi 2015 {published data only}
-
- Besi E, Boniface DR, Cregg R, Zakrzewska JM. Comparison of tolerability and adverse symptoms in oxcarbazepine and carbamazepine in the treatment of trigeminal neuralgia and neuralgiform headaches using the Liverpool Adverse Events Profile (AEP). Journal of Headache and Pain 2015;16:563. - PMC - PubMed
Beydoun 2002 {published data only}
-
- Beydoun A, D'Souza J. Oxcarbazepine versus carbamazepine in patients with new onset trigeminal neuralgia result of double blind comparative trial [abstract no.P06.116]. Neurology 2002;58 Suppl 3:A471.
Beydoun 2007 {published data only}
-
- Beydoun S, Alarcón F, Mangat S, Wan Y. Long‐term safety and tolerability of oxcarbazepine in painful diabetic neuropathy. Acta Neurologica Scandinavica 2007;115(4):284‐8. - PubMed
Brainer‐Lima 2003 {published data only}
-
- Brainer‐Lima P, Naylor F. Oxcarbazepine for cancer‐related neuropathic pain and opiate detoxification. 22nd Annual Meeting of the American Pain Society; 2003 March 20‐23; Chicago (IL). 2003.
Gong 2010 {published data only}
-
- Gong JJ. A randomised controlled trial of 3 drugs for the treatment of idiopathic trigeminal neuralgia [3种药物治疗原发性三叉神经痛的随机对照试验]. Journal of China Traditional Chinese Medicine Information 2010;2(12):74.
Hu 2010 {published data only}
-
- Hu YJ. The comparative efficacy of oxcarbazepine and carbamazepine for trigeminal neuralgia [奥卡西平与卡马西平治疗三次神经痛的疗效对比]. Shandong Medical Journal 2010;50(25):107.
Li 2011 {published data only}
-
- Li G, Zha Q, Chen JZ, Cao QH, Peng WJ. Clinical efficacy of oxcarbazepine for primary trigeminal neuralgia [奥卡西平治疗原发性三叉神经痛临床观察]. Zhejiang Journal of Integrated Traditional Chinese and Western Medicine 2011;21(3):167‐8.
Liebel 2001 {published data only}
-
- Liebel J. Results of a double‐blind trial comparing oxcarbazepine (OXC) vs carbamazepine (CBZ) in newly diagnosed, untreated patients with trigeminal neuralgia. Journal of the Neurological Sciences 2001;187 Suppl 1:S5.
Lindström 1987 {published data only}
-
- Lindström P. The analgesic effect of carbamazepine in trigeminal neuralgia. Pain 1987;28 Suppl 4:S85. - PubMed
Min 2016 {published and unpublished data}
-
- Symptom‐based treatment for neuropathic pain in spinal cord injured patients, randomized clinical trial, 2014. clinicaltrials.gov/ct2/show/study/NCT02180880?term=oxcarbazepine&ran... (accessed 2 October 2015).
-
- Min K, Oh Y, Lee SH, Ryu JS. Symptom‐based treatment of neuropathic pain in spinal cord‐injured patients: a randomized crossover clinical trial. American Journal of Physical Medicine & Rehabilitation 2016;95(5):330‐8. - PubMed
Rémillard 1994 {published data only}
-
- Rémillard G. Oxcarbazepine and intractable trigeminal neuralgia. Epilepsia 1994;35 Suppl 3:S28‐9.
Venancio‐Ramirez 2004 {published data only}
-
- Venancio‐Ramirez L, Hernández‐Santos JR, Tenopala‐Villegas S, Torres‐Huerta JC, Rivera‐León G, Canseco‐Aguilar C. Comparison of oxcarbazepine and gabapentin at standard dose in treatment of pain for postherpetic neuropathy. Revista Mexicana de Anestesiologia 2004;27(3):129‐33.
Zhou 2010 {published data only}
-
- Zhou JF. A clinical research on the pharmacological treatment of trigeminal neuralgia [药物治疗三叉神经痛的临床研究]. Medical Information Operations Sciences Fascicule 2010;1:89.
References to ongoing studies
NCT02219373 {unpublished data only}
-
- NCT02219373. Gabapentin and oxcarbazepine for chronic neuropathic pain in children and adolescents: a clinical effectiveness study. clinicaltrials.gov/ct2/show/NCT02219373 (accessed 15 January 2017).
Additional references
Attal 2006
-
- Attal N, Cruccu G, Haanpää M, Hansson P, Jensen TS, Nurmikko T, et al. EFNS Task Force. EFNS guidelines on pharmacological treatment of neuropathic pain. European Journal of Neurology 2006;13(11):1153‐69. - PubMed
Attal 2010
-
- Attal N, Cruccu G, Baron R, Haanpää M, Hansson P, Jensen TS, et al. European Federation of Neurological Societies. EFNS guidelines on the pharmacological treatment of neuropathic pain: 2009 revision. European Journal of Neurology 2010;17(9):1113‐e88. [DOI: 10.1111/j.1468-1311.2010.02999.x] - DOI - PubMed
Backonja 2003
-
- Backonja M, Glanzman RL. Gabapentin dosing for neuropathic pain: evidence from randomized placebo‐controlled clinical trials. Clinical Therapeutics 2003;25(1):81‐104. - PubMed
Beydoun 2002a
-
- Beydoun A, Schmidt D, Souza J. Oxcarbazepine versus carbamazepine in trigeminal neuralgia: a meta‐analysis of three double‐blind comparative trials. Neurology 2002;58 Suppl 3:A131.
Beydoun 2002b
-
- Beydoun A, Schmidt D, D'Souza J, on behalf of the oxcarbazepine study group. Meta‐analysis of comparative trials of oxcarbazepine versus carbamazepine in trigeminal neuralgia. 21st American Pain Society Annual Meeting; 2002 Mar 14‐17; Baltimore (MD).
Bieri 1990
-
- Bieri D, Reeve RA, Champion GD, Addicoat L, Ziegler JB. The Faces Pain Scale for the self‐assessment of the severity of pain experienced by children: development, initial validation and preliminary investigation for ratio scale properties. Pain 1990;41(2):139‐50. - PubMed
Birse 2012
Callin 2008
-
- Callin S, Bennett MI. Assessment of neuropathic pain. Continuing Education in Anaesthesia, Critical Care and Pain 2008;8(6):210‐3.
Carpenter 2002
-
- Carpenter KJ, Dickenson AH. Molecular aspects of pain research. Pharmacogenomics Journal 2002;2(2):87‐95. - PubMed
Carrazana 2003
-
- Carrazana E, Mikoshiba I. Rationale and evidence for the use of oxcarbazepine in neuropathic pain. Journal of Pain and Symptom Management 2003;25(5 Suppl):S31‐5. - PubMed
Corrigan 2012
Criscuolo 2005
-
- Criscuolo S, Auletta C, Lippi S, Brogi F, Brogi A. Oxcarbazepine monotherapy in postherpetic neuralgia unresponsive to carbamazepine and gabapentin. Acta Neurologica Scandinavica 2005;111(4):229‐32. - PubMed
Dam 1989
-
- Dam M, Ekberg R, Loyning Y, Waltimo O, Jakobsen L. A double‐blind study comparing oxcarbazepine and carbamazepine in patients with newly diagnosed, previously untreated epilepsy. Epilepsy Research 1989;3(1):70‐6. - PubMed
Davis 2004
-
- Davis MP, Walsh D. Epidemiology of cancer pain and factors influencing poor pain control. American Journal of Hospice and Palliative Care 2004;21(2):137‐42. - PubMed
Deeks 2011
-
- Deeks JJ, Higgins JPT, Altman DG. Chapter 9: Analysing data and undertaking meta‐analyses. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Di Stefano 2014
Dickenson 2002
-
- Dickenson AH, Matthews EA, Suzuki R. Neurobiology of neuropathic pain: mode of action of anticonvulsants. European Journal of Pain 2002;6 Suppl A:51‐60. - PubMed
Eisenberg 2007
-
- Eisenberg E, River Y, Shifrin A, Krivoy N. Antiepileptic drugs in the treatment of neuropathic pain. Drugs 2007;67(9):1265‐89. - PubMed
Finnerup 2007
-
- Finnerup NB, Sindrup SH, Jensen TS. Chronic neuropathic pain: mechanisms, drug targets and measurement. Fundamental Clinical Pharmacology 2007;21(1):129‐36. - PubMed
Foley 2003
-
- Foley KM. Opioids and chronic neuropathic pain. New England Journal of Medicine 2003;348(13):1279‐81. - PubMed
Gill 2011
GRADEpro 2008 [Computer program]
-
- GRADE Working Group, McMaster University. GRADEpro. Version 3.2 for Windows. Hamilton (ON): GRADE Working Group, McMaster University, 2008.
Heliovaara 1987
Higgins 2011a
-
- Higgins JPT, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Higgins 2011b
-
- Higgins JPT, Altman DG, Sterne JAC. Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Higgins 2011c
-
- Higgins JPT, Deeks JJ, Altman DG. Chapter 16: Special topics in statistics. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Illing 2013
-
- Illing PT, Vivian JP, Purcell AW, Rossjohn J, McCluskey J. Human leukocyte antigen‐associated drug hypersensitivity. Current Opinion in Immunology 2013;25(1):81‐9. - PubMed
Jensen 2001
-
- Jensen TS, Gottrup H, Sindrup SH. The clinical picture of neuropathic pain. European Journal of Pharmacology 2001;429(1‐3):1‐11. - PubMed
Jensen 2002
-
- Jensen TS. Anticonvulsants in neuropathic pain: rationale and clinical evidence. European Journal of Pain 2002;6 Suppl A:61‐8. - PubMed
Lindstrom 1987
-
- Lindstrom P. The analgesic effect of carbamazepine in trigeminal neuralgia. Pain 1987;30:S85. - PubMed
Magenta 2005
-
- Magenta P, Arghetti S, Palma F, Jann S, Sterlicchio M, Bianconi C, et al. Oxcarbazepine is effective and safe in the treatment of neuropathic pain: pooled analysis of seven clinical studies. Neurological Sciences 2005;26(4):218‐26. - PubMed
Merskey 1994
-
- Merskey H, Bogduk N. Classification of Chronic Pain: Descriptions of Chronic Pain Syndromes and Definitions of Pain Terms. 2nd Edition. Seattle (WA): IASP Press, 1994.
Moore 2009
Moore 2014
Nasreddine 2007
-
- Nasreddine W, Beydoun A. Oxcarbazepine in neuropathic pain. Expert Opinion on Investigational Drugs 2007;16(10):1615‐25. - PubMed
RevMan 2014 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Ryan 2016
-
- Ryan R, Hill S, Cochrane Consumers and Communication Group. How to GRADE the quality of the evidence, Version 1.0, June 2016. cccrg.cochrane.org/author‐resources (accessed prior to 16 November 2017).
Schmader 2002
-
- Schmader KE. Epidemiology and impact on quality of life of postherpetic neuralgia and painful diabetic neuropathy. Clinical Journal of Pain 2002;18(6):350‐4. - PubMed
Schünemann 2011a
-
- Schünemann HJ, Oxman AD, Higgins JPT, Vist GE, Glasziou P, Guyatt GH. Chapter 11: Presenting results and 'Summary of findings' tables. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Schünemann 2011b
-
- Schünemann HJ, Oxman AD, Vist GE, Higgins JPT, Deeks JJ, Glasziou P, et al. Chapter 12: Interpreting results and drawing conclusions. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Suzuki 2004
-
- Suzuki R, Rygh LJ, Dickenson AH. Bad news from the brain: descending 5‐HT pathways that control spinal pain processing trends. Trends in Pharmacological Sciences 2004;25(12):613‐7. - PubMed
Treede 2008
-
- Treede RD, Jensen TS, Campbell JN, Cruccu G, Dostrovsky JO, Griffin JW, et al. Neuropathic pain: redefinition and a grading system for clinical and research purposes. Neurology 2008;70(18):1630‐5. - PubMed
Verma 2005
-
- Verma S, Estanislao L, Simpson D. HIV‐associated neuropathic pain: epidemiology, pathophysiology and management. CNS Drugs 2005;19(4):325‐34. - PubMed
Ware 1992
-
- Ware JE Jr, Sherbourne CD. The MOS 36‐item Short‐Form Health Survey (SF‐36). I. Conceptual framework and item selection. Medical Care 1992;30(6):473‐83. - PubMed
Werhagen 2004
-
- Werhagen L, Budh CN, Hultling C, Molander C. Neuropathic pain after traumatic spinal cord injury? Relations to gender, spinal level, completeness, and age at the time of injury. Spinal Cord 2004;42(12):665‐73. - PubMed
Wiffen 2010
Wiffen 2013
Wiffen 2014
Williamson 2005
-
- Williamson A, Hoggart B. Pain: a review of three commonly used pain rating scales. Journal of Clinical Nursing 2005;14(7):798‐804. - PubMed
References to other published versions of this review
Zhou 2009
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

