More than a biomarker: the systemic consequences of heparan sulfate fragments released during endothelial surface layer degradation (2017 Grover Conference Series)
- PMID: 29199903
- PMCID: PMC5731723
- DOI: 10.1177/2045893217745786
More than a biomarker: the systemic consequences of heparan sulfate fragments released during endothelial surface layer degradation (2017 Grover Conference Series)
Abstract
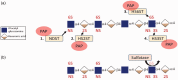
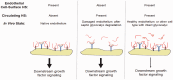
Advances in tissue fixation and imaging techniques have yielded increasing appreciation for the glycosaminoglycan-rich endothelial glycocalyx and its in vivo manifestation, the endothelial surface layer (ESL). Pathological loss of the ESL during critical illness promotes local endothelial dysfunction and, consequently, organ injury. Glycosaminoglycan fragments, such as heparan sulfate, are released into the plasma of animals and humans after ESL degradation and have thus served as a biomarker of endothelial injury. The development of state-of-the-art glycomic techniques, however, has revealed that these circulating heparan sulfate fragments are capable of influencing growth factor and other signaling pathways distant to the site of ESL injury. This review summarizes the current state of knowledge concerning the local (i.e. endothelial injury) and systemic (i.e. para- or endocrine) consequences of ESL degradation and identifies opportunities for future, novel investigations.
Keywords: glycocalyx; glycosaminoglycans; pulmonary endothelium; sepsis/multiple organ failure.
Figures

Similar articles
-
The circulating glycosaminoglycan signature of respiratory failure in critically ill adults.J Biol Chem. 2014 Mar 21;289(12):8194-202. doi: 10.1074/jbc.M113.539452. Epub 2014 Feb 7. J Biol Chem. 2014. PMID: 24509853 Free PMC article. Clinical Trial.
-
Endothelial Heparan Sulfate Proteoglycans in Sepsis: The Role of the Glycocalyx.Semin Thromb Hemost. 2021 Apr;47(3):274-282. doi: 10.1055/s-0041-1725064. Epub 2021 Apr 1. Semin Thromb Hemost. 2021. PMID: 33794552 Review.
-
The Pulmonary Endothelial Glycocalyx in ARDS: A Critical Role for Heparan Sulfate.Curr Top Membr. 2018;82:33-52. doi: 10.1016/bs.ctm.2018.08.005. Epub 2018 Sep 27. Curr Top Membr. 2018. PMID: 30360782
-
Circulating Heparan Sulfate Fragments Attenuate Histone-Induced Lung Injury Independently of Histone Binding.Shock. 2017 Dec;48(6):666-673. doi: 10.1097/SHK.0000000000000907. Shock. 2017. PMID: 28538085 Free PMC article.
-
Degradation of the endothelial glycocalyx in clinical settings: searching for the sheddases.Br J Clin Pharmacol. 2015 Sep;80(3):389-402. doi: 10.1111/bcp.12629. Epub 2015 May 22. Br J Clin Pharmacol. 2015. PMID: 25778676 Free PMC article. Review.
Cited by
-
Thrombin-cleaved syndecan-3/-4 ectodomain fragments mediate endothelial barrier dysfunction.PLoS One. 2019 May 15;14(5):e0214737. doi: 10.1371/journal.pone.0214737. eCollection 2019. PLoS One. 2019. PMID: 31091226 Free PMC article.
-
The influenza-injured lung microenvironment promotes MRSA virulence, contributing to severe secondary bacterial pneumonia.Cell Rep. 2022 Nov 29;41(9):111721. doi: 10.1016/j.celrep.2022.111721. Cell Rep. 2022. PMID: 36450248 Free PMC article.
-
N-Acetylcysteine and Other Sulfur-Donors as a Preventative and Adjunct Therapy for COVID-19.Adv Pharmacol Pharm Sci. 2022 Aug 10;2022:4555490. doi: 10.1155/2022/4555490. eCollection 2022. Adv Pharmacol Pharm Sci. 2022. PMID: 35992575 Free PMC article. Review.
-
VSIG4 interaction with heparan sulfates inhibits VSIG4-complement binding.Glycobiology. 2023 Aug 14;33(7):591-604. doi: 10.1093/glycob/cwad050. Glycobiology. 2023. PMID: 37341346 Free PMC article.
-
The Endothelial Glycocalyx: A Fundamental Determinant of Vascular Permeability in Sepsis.Pediatr Crit Care Med. 2020 May;21(5):e291-e300. doi: 10.1097/PCC.0000000000002266. Pediatr Crit Care Med. 2020. PMID: 32132499 Free PMC article. Review.
References
-
- Luft JH. Fine structures of capillary and endocapillary layer as revealed by ruthenium red. Fed Proc 1966; 25(6): 1773–1783. - PubMed
-
- Schmidt EP, Kuebler WM, Lee WL, et al. Adhesion molecules: master controllers of the circulatory system. Compr Physiol 2016; 6(2): 945–973. - PubMed
-
- Van Teeffelen JW, Brands J, Stroes ES, et al. Endothelial glycocalyx: sweet shield of blood vessels. Trends Cardiovasc Med 2007; 17(3): 101–105. - PubMed
-
- van den Berg BM, Vink H, Spaan JA. The endothelial glycocalyx protects against myocardial edema. Circ Res 2003; 92(6): 592–594. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

