High performance affinity chromatography and related separation methods for the analysis of biological and pharmaceutical agents
- PMID: 29200216
- PMCID: PMC5768458
- DOI: 10.1039/c7an01469d
High performance affinity chromatography and related separation methods for the analysis of biological and pharmaceutical agents
Abstract
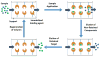
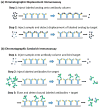
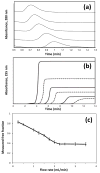
The last few decades have witnessed the development of many high-performance separation methods that use biologically related binding agents. The combination of HPLC with these binding agents results in a technique known as high performance affinity chromatography (HPAC). This review will discuss the general principles of HPAC and related techniques, with an emphasis on their use for the analysis of biological compounds and pharmaceutical agents. Various types of binding agents for these methods will be considered, including antibodies, immunoglobulin-binding proteins, aptamers, enzymes, lectins, transport proteins, lipids, and carbohydrates. Formats that will be discussed for these methods range from the direct detection of an analyte to indirect detection based on chromatographic immunoassays, as well as schemes based on analyte extraction or depletion, post-column detection, and multi-column systems. The use of biological agents in HPLC for chiral separations will also be considered, along with the use of HPAC as a tool to screen or study biological interactions. Various examples will be presented to illustrate these approaches and their applications in fields such as biochemistry, clinical chemistry, and pharmaceutical research.
Figures


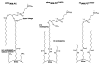
Similar articles
-
Analysis of Biological Interactions by Affinity Chromatography: Clinical and Pharmaceutical Applications.Clin Chem. 2017 Jun;63(6):1083-1093. doi: 10.1373/clinchem.2016.262253. Epub 2017 Apr 10. Clin Chem. 2017. PMID: 28396561 Free PMC article. Review.
-
Pharmaceutical and biomedical applications of affinity chromatography: recent trends and developments.J Pharm Biomed Anal. 2012 Oct;69:93-105. doi: 10.1016/j.jpba.2012.01.004. Epub 2012 Jan 14. J Pharm Biomed Anal. 2012. PMID: 22305083 Free PMC article. Review.
-
Applications of silica supports in affinity chromatography.J Sep Sci. 2006 Apr;29(6):719-37. doi: 10.1002/jssc.200500501. J Sep Sci. 2006. PMID: 16830485 Review.
-
Affinity chromatography: a review of clinical applications.Clin Chem. 1999 May;45(5):593-615. Clin Chem. 1999. PMID: 10222345 Review.
-
Clinical and pharmaceutical applications of affinity ligands in capillary electrophoresis: A review.J Pharm Biomed Anal. 2020 Jan 5;177:112882. doi: 10.1016/j.jpba.2019.112882. Epub 2019 Sep 12. J Pharm Biomed Anal. 2020. PMID: 31542417 Free PMC article. Review.
Cited by
-
Development of a microcolumn one-site immunometric assay for a protein biomarker: Analysis of alpha1-acid glycoprotein.J Chromatogr A. 2020 Jan 11;1610:460558. doi: 10.1016/j.chroma.2019.460558. Epub 2019 Sep 20. J Chromatogr A. 2020. PMID: 31564560 Free PMC article.
-
Route and Type of Formulation Administered Influences the Absorption and Disposition of Vitamin B12 Levels in Serum.J Funct Biomater. 2018 Jan 21;9(1):12. doi: 10.3390/jfb9010012. J Funct Biomater. 2018. PMID: 29361736 Free PMC article.
-
Affinity monolith chromatography: A review of general principles and recent developments.Electrophoresis. 2021 Dec;42(24):2577-2598. doi: 10.1002/elps.202100163. Epub 2021 Aug 12. Electrophoresis. 2021. PMID: 34293192 Free PMC article. Review.
-
Affinity chromatography: A review of trends and developments over the past 50 years.J Chromatogr B Analyt Technol Biomed Life Sci. 2020 Nov 10;1157:122332. doi: 10.1016/j.jchromb.2020.122332. Epub 2020 Aug 14. J Chromatogr B Analyt Technol Biomed Life Sci. 2020. PMID: 32871378 Free PMC article. Review.
-
Review of Eukaryote Cellular Membrane Lipid Composition, with Special Attention to the Fatty Acids.Int J Mol Sci. 2023 Oct 28;24(21):15693. doi: 10.3390/ijms242115693. Int J Mol Sci. 2023. PMID: 37958678 Free PMC article. Review.
References
-
- Pfaunmiller EL, Bas J, Brooks M, Milanuk M, Rodriguez E, Vargas J, Matsuda R, Hage DS. In: Analytical Separation Science. Anderson JL, Berthod A, Pino V, Stalcup AM, editors. WILEY-VCH; Weinheim: 2015. pp. 461–482.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

