Dose-Dependent Rescue of KO Amelogenin Enamel by Transgenes in Vivo
- PMID: 29201008
- PMCID: PMC5696357
- DOI: 10.3389/fphys.2017.00932
Dose-Dependent Rescue of KO Amelogenin Enamel by Transgenes in Vivo
Abstract
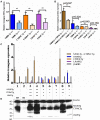
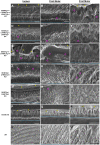
Mice lacking amelogenin (KO) have hypoplastic enamel. Overexpression of the most abundant amelogenin splice variant M180 and LRAP transgenes can substantially improve KO enamel, but only ~40% of the incisor thickness is recovered and the prisms are not as tightly woven as in WT enamel. This implies that the compositional complexity of the enamel matrix is required for different aspects of enamel formation, such as organizational structure and thickness. The question arises, therefore, how important the ratio of different matrix components, and in particular amelogenin splice products, is in enamel formation. Can optimal expression levels of amelogenin transgenes representing both the most abundant splice variants and cleavage product at protein levels similar to that of WT improve the enamel phenotype of KO mice? Addressing this question, our objective was here to understand dosage effects of amelogenin transgenes (Tg) representing the major splice variants M180 and LRAP and cleavage product CTRNC on enamel properties. Amelogenin KO mice were mated with M180Tg, CTRNCTg and LRAPTg mice to generate M180Tg and CTRNCTg double transgene and M180Tg, CTRNCTg, LRAPTg triple transgene mice with transgene hemizygosity (on one allelle) or homozygosity (on both alleles). Transgene homo- vs. hemizygosity was determined by qPCR and relative transgene expression confirmed by Western blot. Enamel volume and mineral density were analyzed by microCT, thickness and structure by SEM, and mechanical properties by Vickers microhardness testing. There were no differences in incisor enamel thickness between amelogenin KO mice with three or two different transgenes, but mice homozygous for a given transgene had significantly thinner enamel than mice hemizygous for the transgene (p < 0.05). The presence of the LRAPTg did not improve the phenotype of M180Tg/CTRNCTg/KO enamel. In the absence of endogenous amelogenin, the addition of amelogenin transgenes representing the most abundant splice variants and cleavage product can rescue abnormal enamel properties and structure, but only up to a maximum of ~80% that of molar and ~40% that of incisor wild-type enamel.
Keywords: amelogenin; enamel development; knockout; mineralization; transgenic.
Figures


References
-
- Aldred M. J., Crawford P. J., Roberts E., Thomas N. S. (1992). Identification of a nonsense mutation in the amelogenin gene (AMELX) in a family with X-linked amelogenesis imperfecta (AIH1). Hum. Genet. 90, 413–416. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

