Membrane Dynamics and Multiple Functions of Oil Bodies in Seeds and Leaves
- PMID: 29203559
- PMCID: PMC5761825
- DOI: 10.1104/pp.17.01522
Membrane Dynamics and Multiple Functions of Oil Bodies in Seeds and Leaves
Abstract
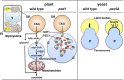
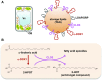
Oil bodies have multiple functions: oleosin-mediated freezing tolerance of seeds, direct interaction with glyoxysomes for lipid degradation in seedlings, and antifungal compound production in leaves.
Figures





Similar articles
-
Leaf oil bodies are subcellular factories producing antifungal oxylipins.Curr Opin Plant Biol. 2015 Jun;25:145-50. doi: 10.1016/j.pbi.2015.05.019. Epub 2015 Jun 5. Curr Opin Plant Biol. 2015. PMID: 26051035 Review.
-
Sterols are required for the coordinated assembly of lipid droplets in developing seeds.Nat Commun. 2021 Sep 22;12(1):5598. doi: 10.1038/s41467-021-25908-6. Nat Commun. 2021. PMID: 34552075 Free PMC article.
-
Lipid Droplets: Packing Hydrophobic Molecules Within the Aqueous Cytoplasm.Annu Rev Plant Biol. 2023 May 22;74:195-223. doi: 10.1146/annurev-arplant-070122-021752. Epub 2022 Nov 22. Annu Rev Plant Biol. 2023. PMID: 36413579 Review.
-
Arabidopsis lipid droplet-associated protein (LDAP) - interacting protein (LDIP) influences lipid droplet size and neutral lipid homeostasis in both leaves and seeds.Plant J. 2017 Dec;92(6):1182-1201. doi: 10.1111/tpj.13754. Epub 2017 Nov 27. Plant J. 2017. PMID: 29083105
-
Arabidopsis SEIPIN Proteins Modulate Triacylglycerol Accumulation and Influence Lipid Droplet Proliferation.Plant Cell. 2015 Sep;27(9):2616-36. doi: 10.1105/tpc.15.00588. Epub 2015 Sep 11. Plant Cell. 2015. PMID: 26362606 Free PMC article.
Cited by
-
Lipid Droplet Isolation from Arabidopsis thaliana Leaves.Bio Protoc. 2020 Dec 20;10(24):e3867. doi: 10.21769/BioProtoc.3867. eCollection 2020 Dec 20. Bio Protoc. 2020. PMID: 33659507 Free PMC article.
-
Bacillus subtilis biofilm matrix components target seed oil bodies to promote growth and anti-fungal resistance in melon.Nat Microbiol. 2022 Jul;7(7):1001-1015. doi: 10.1038/s41564-022-01134-8. Epub 2022 Jun 6. Nat Microbiol. 2022. PMID: 35668112 Free PMC article.
-
Seeds as oil factories.Plant Reprod. 2018 Sep;31(3):213-235. doi: 10.1007/s00497-018-0325-6. Epub 2018 Feb 10. Plant Reprod. 2018. PMID: 29429143 Review.
-
Polyamine depletion enhances oil body mobilization through possible regulation of oleosin degradation and aquaporin abundance on its membrane.Plant Signal Behav. 2023 Dec 31;18(1):2217027. doi: 10.1080/15592324.2023.2217027. Plant Signal Behav. 2023. PMID: 37243675 Free PMC article.
-
Plant Unsaturated Fatty Acids: Multiple Roles in Stress Response.Front Plant Sci. 2020 Sep 4;11:562785. doi: 10.3389/fpls.2020.562785. eCollection 2020. Front Plant Sci. 2020. PMID: 33013981 Free PMC article. Review.
References
-
- Abell BM, Hahn M, Holbrook LA, Moloney MM (2004) Membrane topology and sequence requirements for oil body targeting of oleosin. Plant J 37: 461–470 - PubMed
-
- Abell BM, High S, Moloney MM (2002) Membrane protein topology of oleosin is constrained by its long hydrophobic domain. J Biol Chem 277: 8602–8610 - PubMed
-
- Avila CA, Arevalo-Soliz LM, Lorence A, Goggin FL (2013) Expression of α-DIOXYGENASE 1 in tomato and Arabidopsis contributes to plant defenses against aphids. Mol Plant Microbe Interact 26: 977–986 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

