Epigenetic therapy activates type I interferon signaling in murine ovarian cancer to reduce immunosuppression and tumor burden
- PMID: 29203668
- PMCID: PMC5754782
- DOI: 10.1073/pnas.1712514114
Epigenetic therapy activates type I interferon signaling in murine ovarian cancer to reduce immunosuppression and tumor burden
Abstract
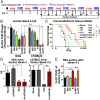
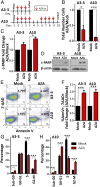
Ovarian cancer is the most lethal of all gynecological cancers, and there is an urgent unmet need to develop new therapies. Epithelial ovarian cancer (EOC) is characterized by an immune suppressive microenvironment, and response of ovarian cancers to immune therapies has thus far been disappointing. We now find, in a mouse model of EOC, that clinically relevant doses of DNA methyltransferase and histone deacetylase inhibitors (DNMTi and HDACi, respectively) reduce the immune suppressive microenvironment through type I IFN signaling and improve response to immune checkpoint therapy. These data indicate that the type I IFN response is required for effective in vivo antitumorigenic actions of the DNMTi 5-azacytidine (AZA). Through type I IFN signaling, AZA increases the numbers of CD45+ immune cells and the percentage of active CD8+ T and natural killer (NK) cells in the tumor microenvironment, while reducing tumor burden and extending survival. AZA also increases viral defense gene expression in both tumor and immune cells, and reduces the percentage of macrophages and myeloid-derived suppressor cells in the tumor microenvironment. The addition of an HDACi to AZA enhances the modulation of the immune microenvironment, specifically increasing T and NK cell activation and reducing macrophages over AZA treatment alone, while further increasing the survival of the mice. Finally, a triple combination of DNMTi/HDACi plus the immune checkpoint inhibitor α-PD-1 provides the best antitumor effect and longest overall survival, and may be an attractive candidate for future clinical trials in ovarian cancer.
Keywords: 5-azacytidine; histone deacetylase inhibitors; immunosuppression; ovarian cancer; type I interferon.
Conflict of interest statement
Conflict of interest statement: S.B.B. and C.A.Z. have a collaborative research agreement with Janssen. V.B., K.R.W., G.S.C., and K.E.B. are employed by Janssen.
Figures
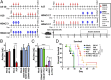

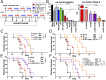
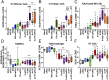
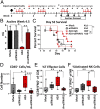
Comment in
-
Reply to Haffner et al.: DNA hypomethylation renders tumors more immunogenic.Proc Natl Acad Sci U S A. 2018 Sep 11;115(37):E8583-E8584. doi: 10.1073/pnas.1811015115. Epub 2018 Sep 4. Proc Natl Acad Sci U S A. 2018. PMID: 30181297 Free PMC article. No abstract available.
-
Hypomethylation, endogenous retrovirus expression, and interferon signaling in testicular germ cell tumors.Proc Natl Acad Sci U S A. 2018 Sep 11;115(37):E8580-E8582. doi: 10.1073/pnas.1803292115. Epub 2018 Sep 4. Proc Natl Acad Sci U S A. 2018. PMID: 30181298 Free PMC article. No abstract available.
References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7–30. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

