Developmental HSC Microenvironments: Lessons from Zebrafish
- PMID: 29204828
- PMCID: PMC5953202
- DOI: 10.1007/978-3-319-69194-7_4
Developmental HSC Microenvironments: Lessons from Zebrafish
Abstract
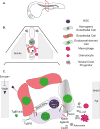
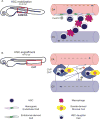
Hematopoietic stem cells (HSCs) posses the ability to maintain the blood system of an organism from birth to adulthood. The behavior of HSCs is modulated by its microenvironment. During development, HSCs acquire the instructions to self-renew and differentiate into all blood cell fates by passing through several developmental microenvironments. In this chapter, we discuss the signals and cell types that inform HSC decisions throughout ontogeny with a focus on HSC specification, mobilization, migration, and engraftment.
Keywords: Blood development; Hematopoietic niche; Hematopoietic stem cell; Zebrafish.
Figures

Similar articles
-
Mobilization and homing of hematopoietic stem cells.Adv Exp Med Biol. 2012;741:152-70. doi: 10.1007/978-1-4614-2098-9_11. Adv Exp Med Biol. 2012. PMID: 22457109 Review.
-
Hematopoietic stem cell development: using the zebrafish to identify the signaling networks and physical forces regulating hematopoiesis.Methods Cell Biol. 2011;105:117-36. doi: 10.1016/B978-0-12-381320-6.00005-9. Methods Cell Biol. 2011. PMID: 21951528
-
tfec controls the hematopoietic stem cell vascular niche during zebrafish embryogenesis.Blood. 2016 Sep 8;128(10):1336-45. doi: 10.1182/blood-2016-04-710137. Epub 2016 Jul 11. Blood. 2016. PMID: 27402973
-
Regulation of hematopoietic stem cells in the niche.Sci China Life Sci. 2015 Dec;58(12):1209-15. doi: 10.1007/s11427-015-4960-y. Epub 2015 Nov 13. Sci China Life Sci. 2015. PMID: 26563156 Review.
-
Analyzing hematopoietic stem cell homing, lodgment, and engraftment to better understand the bone marrow niche.Ann N Y Acad Sci. 2014 Mar;1310:119-28. doi: 10.1111/nyas.12329. Epub 2014 Jan 15. Ann N Y Acad Sci. 2014. PMID: 24428368 Review.
Cited by
-
Dhx15 regulates zebrafish definitive hematopoiesis through the unfolded protein response pathway.Cancer Sci. 2021 Sep;112(9):3884-3894. doi: 10.1111/cas.15002. Epub 2021 Jul 11. Cancer Sci. 2021. PMID: 34077586 Free PMC article.
-
Inflammation in Development and Aging: Insights from the Zebrafish Model.Int J Mol Sci. 2024 Feb 10;25(4):2145. doi: 10.3390/ijms25042145. Int J Mol Sci. 2024. PMID: 38396822 Free PMC article. Review.
-
Excessive R-loops trigger an inflammatory cascade leading to increased HSPC production.Dev Cell. 2021 Mar 8;56(5):627-640.e5. doi: 10.1016/j.devcel.2021.02.006. Epub 2021 Mar 1. Dev Cell. 2021. PMID: 33651979 Free PMC article.
-
Splicing factor deficits render hematopoietic stem and progenitor cells sensitive to STAT3 inhibition.Cell Rep. 2022 Dec 13;41(11):111825. doi: 10.1016/j.celrep.2022.111825. Cell Rep. 2022. PMID: 36516770 Free PMC article.
-
Bloody Zebrafish: Novel Methods in Normal and Malignant Hematopoiesis.Front Cell Dev Biol. 2018 Oct 15;6:124. doi: 10.3389/fcell.2018.00124. eCollection 2018. Front Cell Dev Biol. 2018. PMID: 30374440 Free PMC article.
References
-
- Artavanis-Tsakonas S, Rand MD, Lake RJ. Notch signaling: cell fate control and signal integration in development. Science. 1999;284:770–776. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

