Young plasma reverses age-dependent alterations in hepatic function through the restoration of autophagy
- PMID: 29210183
- PMCID: PMC5770779
- DOI: 10.1111/acel.12708
Young plasma reverses age-dependent alterations in hepatic function through the restoration of autophagy
Abstract
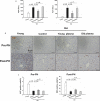
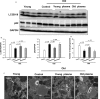
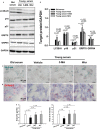
Recent studies showing the therapeutic effect of young blood on aging-associated deterioration of organs point to young blood as the solution for clinical problems related to old age. Given that defective autophagy has been implicated in aging and aging-associated organ injuries, this study was designed to determine the effect of young blood on aging-induced alterations in hepatic function and underlying mechanisms, with a focus on autophagy. Aged rats (22 months) were treated with pooled plasma (1 ml, intravenously) collected from young (3 months) or aged rats three times per week for 4 weeks, and 3-methyladenine or wortmannin was used to inhibit young blood-induced autophagy. Aging was associated with elevated levels of alanine transaminase and aspartate aminotransferase, lipofuscin accumulation, steatosis, fibrosis, and defective liver regeneration after partial hepatectomy, which were significantly attenuated by young plasma injections. Young plasma could also restore aging-impaired autophagy activity. Inhibition of the young plasma-restored autophagic activity abrogated the beneficial effect of young plasma against hepatic injury with aging. In vitro, young serum could protect old hepatocytes from senescence, and the antisenescence effect of young serum was abrogated by 3-methyladenine, wortmannin, or small interfering RNA to autophagy-related protein 7. Collectively, our data indicate that young plasma could ameliorate age-dependent alterations in hepatic function partially via the restoration of autophagy.
Keywords: aging; autophagy; hepatocyte; liver injury; liver regeneration; rat; senescence; young blood.
© 2017 The Authors. Aging Cell published by the Anatomical Society and John Wiley & Sons Ltd.
Figures



References
-
- Baker, D. J. , Wijshake, T. , Tchkonia, T. , LeBrasseur, N. K. , Childs, B. G. , van de Sluis, B. , … van Deursen, J. M. (2011). Clearance of p16Ink4a‐positive senescent cells delays ageing‐associated disorders. Nature, 479, 232–236. https://doi.org/10.1038/nature10600 - DOI - PMC - PubMed
-
- Campisi, J. (2005). Senescent cells, tumor suppression, and organismal aging: Good citizens, bad neighbors. Cell, 120, 513–522. https://doi.org/10.1016/j.cell.2005.02.003 - DOI - PubMed
-
- Campisi, J. , & d'Adda, DF (2007). Cellular senescence: When bad things happen to good cells. Nature Reviews Molecular Cell Biology, 8, 729–740. https://doi.org/10.1038/nrm2233 - DOI - PubMed
-
- Chang, J. , Wang, Y. , Shao, L. , Laberge, R. M. , Demaria, M. , Campisi, J. , … Zhou, D. (2016). Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice. Nature Medicine, 22, 78–83. https://doi.org/10.1038/nm.4010 - DOI - PMC - PubMed
-
- Conboy, I. M. , Conboy, M. J. , Wagers, A. J. , Girma, E. R. , Weissman, I. L. , & Rando, T. A. (2005). Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature, 433, 760–764. https://doi.org/10.1038/nature03260 - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

