Neurochemical and Behavior Deficits in Rats with Iron and Rotenone Co-treatment: Role of Redox Imbalance and Neuroprotection by Biochanin A
- PMID: 29217997
- PMCID: PMC5703859
- DOI: 10.3389/fnins.2017.00657
Neurochemical and Behavior Deficits in Rats with Iron and Rotenone Co-treatment: Role of Redox Imbalance and Neuroprotection by Biochanin A
Abstract
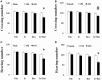
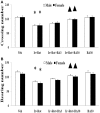
Increasing evidences show that the etiology of Parkinson's disease (PD) is multifactorial. Studying the combined effect of several factors is becoming a hot topic in PD research. On one hand, iron is one of the essential trace metals for human body; on the other hand, iron may be involved in the etiopathogenesis of PD. In our present study, the rats with increased neonatal iron (120 μg/g bodyweight) supplementation were treated with rotenone (0.5 mg/kg) when they were aged to 14 weeks. We observed that iron and rotenone co-treatment induced significant behavior deficits (time-dependent) and striatal dopamine depletion in the male and female rats, while they did not do so when they were used alone. No significant change in striatal 5-hydroxytryptamine content was observed in the male and female rats with iron and rotenone co-treatment. Also, iron and rotenone co-treatment significantly decreased substantia nigra TH expression in the male rats. Furthermore, co-treatment with iron and rotenone significantly induced malondialdehyde increase and glutathione decrease in the substantia nigra of male and female rats. There was no significant change in cerebellar malondialdehyde and glutathione content of the rats co-treated with iron and rotenone. Interestingly, biochanin A significantly attenuated striatal dopamine depletion and improved behavior deficits (dose-dependently) in the male and female rats with iron and rotenone co-treatment. Biochanin A treatment also significantly alleviated substantia nigra TH expression reduction in the male rats co-treated with iron and rotenone. Finally, biochanin A significantly decreased malondialdehyde content and increased glutathione content in the substantia nigra of male and female rats with iron and rotenone co-treatment. Our results indicate that iron and rotenone co-treatment may result in aggravated neurochemical and behavior deficits through inducing redox imbalance and increased neonatal iron supplementation may participate in the etiopathogenesis of PD. Moreover, biochanin A may exert dopaminergic neuroprotection by maintaining redox balance.
Keywords: biochanin A; iron; parkinson's disease; redox balance; roteone.
Figures







Similar articles
-
Aging-related rotenone-induced neurochemical and behavioral deficits: role of SIRT2 and redox imbalance, and neuroprotection by AK-7.Drug Des Devel Ther. 2015 May 7;9:2553-63. doi: 10.2147/DDDT.S81539. eCollection 2015. Drug Des Devel Ther. 2015. PMID: 26089639 Free PMC article.
-
Behavioral and Neurochemical Deficits in Aging Rats with Increased Neonatal Iron Intake: Silibinin's Neuroprotection by Maintaining Redox Balance.Front Aging Neurosci. 2015 Oct 28;7:206. doi: 10.3389/fnagi.2015.00206. eCollection 2015. Front Aging Neurosci. 2015. PMID: 26578951 Free PMC article.
-
Aggravated behavioral and neurochemical deficits and redox imbalance in mice with enhanced neonatal iron intake: improvement by biochanin A and role of microglial p38 activation.Nutr Neurosci. 2021 Mar;24(3):161-172. doi: 10.1080/1028415X.2019.1611021. Epub 2019 May 3. Nutr Neurosci. 2021. PMID: 31050314
-
Nicotine-Induced Neuroprotection in Rotenone In Vivo and In Vitro Models of Parkinson's Disease: Evidences for the Involvement of the Labile Iron Pool Level as the Underlying Mechanism.Neurotox Res. 2019 Jan;35(1):71-82. doi: 10.1007/s12640-018-9931-1. Epub 2018 Jul 13. Neurotox Res. 2019. PMID: 30006684
-
Acute intranigral infusion of rotenone in rats causes progressive biochemical lesions in the striatum similar to Parkinson's disease.Brain Res. 2005 Jul 12;1049(2):147-55. doi: 10.1016/j.brainres.2005.04.051. Brain Res. 2005. PMID: 15936733
Cited by
-
Behavioral Tests in Neurotoxin-Induced Animal Models of Parkinson's Disease.Antioxidants (Basel). 2020 Oct 16;9(10):1007. doi: 10.3390/antiox9101007. Antioxidants (Basel). 2020. PMID: 33081318 Free PMC article. Review.
-
Mechanisms Behind the Pharmacological Application of Biochanin-A: A review.F1000Res. 2023 Dec 11;12:107. doi: 10.12688/f1000research.126059.3. eCollection 2023. F1000Res. 2023. PMID: 38106650 Free PMC article. Review.
-
Amelioration of Mitochondrial Quality Control and Proteostasis by Natural Compounds in Parkinson's Disease Models.Int J Mol Sci. 2019 Oct 21;20(20):5208. doi: 10.3390/ijms20205208. Int J Mol Sci. 2019. PMID: 31640129 Free PMC article. Review.
-
Using mitochondrial respiration inhibitors to design a novel model of bipolar disorder-like phenotype with construct, face and predictive validity.Transl Psychiatry. 2021 Feb 12;11(1):123. doi: 10.1038/s41398-021-01215-y. Transl Psychiatry. 2021. PMID: 33579900 Free PMC article.
-
Neuroprotective potential of biochanin-A and review of the molecular mechanisms involved.Mol Biol Rep. 2023 Jun;50(6):5369-5378. doi: 10.1007/s11033-023-08397-2. Epub 2023 Apr 11. Mol Biol Rep. 2023. PMID: 37039995 Review.
References
-
- Baldereschi M., Di Carlo A., Rocca W. A., Vanni P., Maggi S., Perissinotto E., et al. . (2000). Parkinson's disease and parkinsonism in a longitudinal study: two-fold higher incidence in men. ILSA working group. Italian longitudinal study on aging. Neurology 55, 1358–1363. 10.1212/WNL.55.9.1358 - DOI - PubMed
-
- Beard J. (2003). Iron deficiency alters brain development and functioning. J. Nutr. 133, 1468S−1472S. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

