Structural Dynamics of the MscL C-terminal Domain
- PMID: 29222414
- PMCID: PMC5722894
- DOI: 10.1038/s41598-017-17396-w
Structural Dynamics of the MscL C-terminal Domain
Abstract
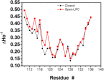
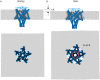
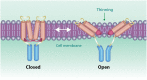
The large conductance mechanosensitive channel (MscL), acts as an osmoprotective emergency valve in bacteria by opening a large, water-filled pore in response to changes in membrane tension. In its closed configuration, the last 36 residues at the C-terminus form a bundle of five α-helices co-linear with the five-fold axis of symmetry. Here, we examined the structural dynamics of the C-terminus of EcMscL using site-directed spin labelling electron paramagnetic resonance (SDSL EPR) spectroscopy. These experiments were complemented with computational modelling including molecular dynamics (MD) simulations and finite element (FE) modelling. Our results show that under physiological conditions, the C-terminus is indeed an α-helical bundle, located near the five-fold symmetry axis of the molecule. Both experiments and computational modelling demonstrate that only the top part of the C-terminal domain (from the residue A110 to E118) dissociates during the channel gating, while the rest of the C-terminus stays assembled. This result is consistent with the view that the C-terminus functions as a molecular sieve and stabilizer of the oligomeric MscL structure as previously suggested.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures





References
-
- Cox, C. D., Bavi, N. & Martinac, B. Bacterial Mechanosensors. Annual Review of Physiology80 (2017). - PubMed
-
- Cox, C., Bavi, N. & Martinac, B. Origin of the Force: The Force-From-Lipids Principle Applied to Piezo Channels. Current Topics in Membranes (2016). - PubMed
-
- Shabsigh A, et al. Rapid reduction in blood flow to the rat ventral prostate gland after castration: preliminary evidence that androgens influence prostate size by regulating blood flow to the prostate gland and prostatic endothelial cell survival. Prostate. 1998;36:201–206. doi: 10.1002/(SICI)1097-0045(19980801)36:3<201::AID-PROS9>3.0.CO;2-J. - DOI - PubMed
-
- Steinbacher, S., Bass, R., Strop, P., and Rees, D.C. in Mechanosensitive Ion Channels, Part A (ed O. P. Hamill) 1–24 (Elsevier Academic Press, Inc., 2007).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

