Stress-induced Cdk5 activity enhances cytoprotective basal autophagy in Drosophila melanogaster by phosphorylating acinus at serine437
- PMID: 29227247
- PMCID: PMC5760206
- DOI: 10.7554/eLife.30760
Stress-induced Cdk5 activity enhances cytoprotective basal autophagy in Drosophila melanogaster by phosphorylating acinus at serine437
Erratum in
-
Correction: Stress-induced Cdk5 activity enhances cytoprotective basal autophagy in Drosophila melanogaster by phosphorylating acinus at serine437.Elife. 2025 Jul 9;14:e108230. doi: 10.7554/eLife.108230. Elife. 2025. PMID: 40632135 Free PMC article.
Abstract
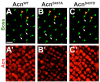
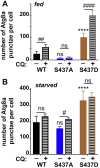
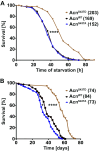
Cdk5 is a post-mitotic kinase with complex roles in maintaining neuronal health. The various mechanisms by which Cdk5 inhibits and promotes neurodegeneration are still poorly understood. Here, we show that in Drosophila melanogaster Cdk5 regulates basal autophagy, a key mechanism suppressing neurodegeneration. In a targeted screen, Cdk5 genetically interacted with Acinus (Acn), a primarily nuclear protein, which promotes starvation-independent, basal autophagy. Loss of Cdk5, or its required cofactor p35, reduces S437-Acn phosphorylation, whereas Cdk5 gain-of-function increases pS437-Acn levels. The phospho-mimetic S437D mutation stabilizes Acn and promotes basal autophagy. In p35 mutants, basal autophagy and lifespan are reduced, but restored to near wild-type levels in the presence of stabilized AcnS437D. Expression of aggregation-prone polyQ-containing proteins or the Amyloid-β42 peptide, but not alpha-Synuclein, enhances Cdk5-dependent phosphorylation of S437-Acn. Our data indicate that Cdk5 is required to maintain the protective role of basal autophagy in the initial responses to a subset of neurodegenerative challenges.
Keywords: D. melanogaster; Drosophila; autophagy; cell biology; compound eye; fat body; neurodegeneration.
Plain language summary
Cells have a problem that we recognize from our own homes: if nobody cleans up, garbage accumulates. Unwanted material in cells can include proteins that clump together and can no longer carry out their normal tasks. If left to build up, these protein aggregates can damage the cell and even kill it. Many neurodegenerative disorders, including Huntington’s disease and Alzheimer's disease, arise when such faulty proteins accumulate inside brain cells. Autophagy is a process that can destroy protein aggregates and other defective material to keep cells healthy. Understanding how cells regulate autophagy is thus of great interest to scientists. A protein called Acinus promotes autophagy and is found in many organisms including fruit flies and humans. All Acinus proteins share a common feature; they contain a site called Serine437 that can be modified by the attachment of a phosphate group, in a process known as phosphorylation. However, the significance of this modification was not clear. Nandi et al. have now asked if this phosphorylation event is important for the role of Acinus in autophagy. The experiments were carried out in the fruit fly, Drosophila melanogaster. Flies were engineered such that the normal Acinus protein was replaced with a mutant version that mimics the phosphorylation at Serine437. These mutant flies had higher levels of Acinus, showed more autophagy, and lived longer when compared to normal flies. Further work identified a protein called Cdk5 as being responsible for attaching phosphate to Acinus at Serine437. Making Cdk5 inactive using experimental tools led to lower levels of autophagy in brain cells and shortened the flies’ life span. Moreover, some aggregation-prone proteins linked to neurodegenerative diseases can enhance the activity of Cdk5 towards Acinus, thereby reducing their own accumulation through elevated autophagy. Together these findings show that phosphorylation of Acinus by Cdk5 maintains healthy brain cells and improves life span by enhancing autophagy. The next step is to understand how phosphorylation at Serine437 stabilizes Acinus to boost autophagy. This may lead to new ways to adjust the levels of autophagy to benefit different organisms.
Conflict of interest statement
No competing interests declared.
Figures


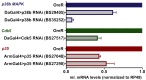
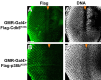

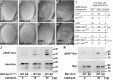
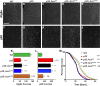
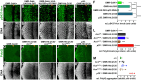
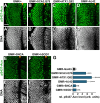
Comment in
-
Cdk5-mediated Acn/Acinus phosphorylation regulates basal autophagy independently of metabolic stress.Autophagy. 2018;14(7):1271-1272. doi: 10.1080/15548627.2018.1441472. Epub 2018 May 21. Autophagy. 2018. PMID: 29782227 Free PMC article.
Similar articles
-
A phosphoswitch at acinus-serine437 controls autophagic responses to cadmium exposure and neurodegenerative stress.Elife. 2022 Jan 17;11:e72169. doi: 10.7554/eLife.72169. Elife. 2022. PMID: 35037620 Free PMC article.
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Drosophila seminal sex peptide associates with rival as well as own sperm, providing SP function in polyandrous females.Elife. 2020 Jul 16;9:e58322. doi: 10.7554/eLife.58322. Elife. 2020. PMID: 32672537 Free PMC article.
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
Cited by
-
Protein modification regulated autophagy in Bombyx mori and Drosophila melanogaster.Front Physiol. 2023 Nov 9;14:1281555. doi: 10.3389/fphys.2023.1281555. eCollection 2023. Front Physiol. 2023. PMID: 38028759 Free PMC article. Review.
-
Manipulating mitochondrial reactive oxygen species alters survival in unexpected ways in a Drosophila Cdk5 model of neurodegeneration.Biol Open. 2024 Jul 15;13(10):bio060515. doi: 10.1242/bio.060515. Epub 2024 Oct 14. Biol Open. 2024. PMID: 39292114 Free PMC article.
-
MANIPULATING MITOCHONDRIAL REACTIVE OXYGEN SPECIES ALTERS SURVIVAL IN UNEXPECTED WAYS IN A DROSOPHILA MODEL OF NEURODEGENERATION.bioRxiv [Preprint]. 2024 Mar 29:2024.03.25.586603. doi: 10.1101/2024.03.25.586603. bioRxiv. 2024. Update in: Biol Open. 2024 Jul 15;13(10):bio060515. doi: 10.1242/bio.060515. PMID: 38585927 Free PMC article. Updated. Preprint.
-
Hyperactive Innate Immunity Causes Degeneration of Dopamine Neurons upon Altering Activity of Cdk5.Cell Rep. 2019 Jan 2;26(1):131-144.e4. doi: 10.1016/j.celrep.2018.12.025. Cell Rep. 2019. PMID: 30605670 Free PMC article.
-
Visual impairment cell non-autonomously dysregulates brain-wide proteostasis.bioRxiv [Preprint]. 2023 Oct 23:2023.10.19.563166. doi: 10.1101/2023.10.19.563166. bioRxiv. 2023. PMID: 37961457 Free PMC article. Preprint.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

