Stability of plant virus-based nanocarriers in gastrointestinal fluids
- PMID: 29231944
- PMCID: PMC5804478
- DOI: 10.1039/c7nr07182e
Stability of plant virus-based nanocarriers in gastrointestinal fluids
Abstract
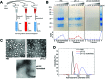
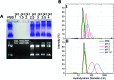
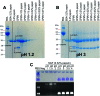
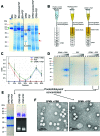
Cowpea mosaic virus (CPMV) is a plant virus which is being extensively investigated as a drug delivery and vaccine nanocarrier for parenteral administration. However, to date little is known about the suitability of plant-based nanocarriers for oral delivery. In this study, the colloidal (i.e. aggregation), physical (i.e. denaturation) and chemical (i.e. digestion of the polypeptides) stability of CPMV and its empty virus-like particles (eVLPs) in conditions resembling the gastrointestinal fluids were evaluated. The nanoparticles were incubated in various simulated gastric and intestinal fluids and in pig gastric and intestinal fluids. CPMV and eVLPs had similar stabilities. In simulated gastric media, they were stable at pH ≥ 2.5. At lower pH destabilisation of the particle structure occurred, which, in turn, rendered the polypeptides extremely sensitive to pepsin digestion. However, both CPMV and eVLPs were stable in simulated intestinal fluids, in pig gastric fluids and in pig intestinal fluids. Thus CPMV, despite being a protein-based nanoparticle, was much more resistant to the harsh GI conditions than soluble proteins. Remarkably, both CPMV and eVLPs incubated in pig gastric and intestinal fluids were not subject to protein adsorption, with no formation of a detectable protein corona. The lack of a protein corona on CPMV and eVLP surfaces in GI fluids would imply that, if orally administered, these nanoparticles could maintain their native surface characteristics; thus, their biological interactions would remain predictable and unchanged. In summary, CPMV and eVLPs can be considered promising nanocarriers for applications requiring oral delivery, given their chemical, physical and colloidal stability and lack of protein adsorption from the environment in most of the tested conditions.
Figures

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

