Retargeting Lentiviruses via SpyCatcher-SpyTag Chemistry for Gene Delivery into Specific Cell Types
- PMID: 29233896
- PMCID: PMC5727413
- DOI: 10.1128/mBio.01860-17
Retargeting Lentiviruses via SpyCatcher-SpyTag Chemistry for Gene Delivery into Specific Cell Types
Abstract
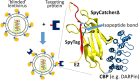
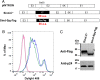
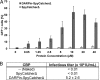
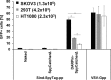
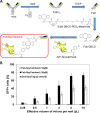
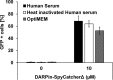
We report a simple strategy for the creation of lentiviral vectors specific to any desired target cells. SpyTag is inserted into an engineered Sindbis virus envelope protein and displayed on the lentivirus surface to create Sindbis virus-SpyTag pseudoparticles (Sind-SpyTag-pp). The SpyTag serves as the covalent anchoring site for a target-cell-specific cell-binding protein (CBP) that is fused to a truncated SpyCatcher (SpyCatcherΔ). Target-cell-specific lentiviruses are created by mixing the Sind-SpyTag-pp and CBP-SpyCatcherΔ in vitro We first used a HER2-binding designed ankyrin repeat protein (DARPin.9.26) as the model CBP. The DARPin-conjugated lentivirus transduced HER2+ SKOV3 cells with an infectious titer of 5.2 × 106 IU/ml, >500-fold higher than the unfunctionalized "naked" virions (<104 IU/ml). The ability of the DARPin-conjugated lentivirus to transduce HER2+ cells correlated with the surface expression level of HER2. Furthermore, these lentiviruses preferentially transduced HER2+ cells in cocultures containing HER2+ and HER2- cells. To enable the use of commercially available monoclonal antibodies (MAbs) as the CBP, we developed a convenient click chemistry-based approach to conjugate MAb-derived Fab fragments to a variant SpyCatcherΔ protein containing a nonnatural amino acid, 4-azido-l-phenylalanine (AzF). Using the HER2-binding trastuzumab as a model cell-specific MAb, we created Fab-conjugated lentiviral vectors that transduced HER2+ SKOV3 cells with an infectious titer of 2.8 × 106 IU/ml, on par with the result achieved using the DARPin-SpyCatcherΔ fusion protein. The ability to create cell-specific lentiviral vectors through chemical conjugation of a CBP should make this approach generalizable to any antibody, giving it broad utility for a wide range of research and clinical applications.IMPORTANCE Lentiviral vectors hold great potential in gene therapy. However, it remains a major hurdle to robustly engineer cell-specific lentiviral vectors. This article reports a simple and effective strategy to functionalize lentiviral vectors with cell-binding proteins, thus retargeting these viruses to cells expressing the binding partner of the CBP. The CBP is genetically or chemically linked to the SpyCatcher. The SpyTag is displayed on the virion surface as a fusion to an engineered Sindbis virus envelope protein and is used as the anchorage site for SpyCatcher-linked CBP. Using this strategy, we created lentiviral vectors highly infectious toward HER2+ cancer cells. The ability to rapidly create cell-specific lentiviral vectors targeting a wide range of cell types should accelerate the development of custom lentiviral vectors for many research and clinical applications.
Keywords: antibody; breast cancer; gene therapy; redirect; specificity.
Copyright © 2017 Kasaraneni et al.
Figures


Similar articles
-
A simple strategy for retargeting lentiviral vectors to desired cell types via a disulfide-bond-forming protein-peptide pair.Sci Rep. 2018 Jul 20;8(1):10990. doi: 10.1038/s41598-018-29253-5. Sci Rep. 2018. PMID: 30030466 Free PMC article.
-
Design and construction of a recombinant lentiviral vector with specific tropism to human epidermal growth factor-overexpressed cancer cells: Developing a new retargeting system for lentivirus vectors.J Gene Med. 2019 Jun;21(6):e3095. doi: 10.1002/jgm.3095. Epub 2019 May 20. J Gene Med. 2019. PMID: 31050357
-
Cell-specific targeting of lentiviral vectors mediated by fusion proteins derived from Sindbis virus, vesicular stomatitis virus, or avian sarcoma/leukosis virus.Retrovirology. 2010 Jan 25;7:3. doi: 10.1186/1742-4690-7-3. Retrovirology. 2010. PMID: 20100344 Free PMC article.
-
Pseudotyped Lentiviral Vectors: One Vector, Many Guises.Hum Gene Ther Methods. 2017 Dec;28(6):291-301. doi: 10.1089/hgtb.2017.084. Epub 2017 Sep 4. Hum Gene Ther Methods. 2017. PMID: 28870117 Review.
-
Pseudotyping Lentiviral Vectors: When the Clothes Make the Virus.Viruses. 2020 Nov 16;12(11):1311. doi: 10.3390/v12111311. Viruses. 2020. PMID: 33207797 Free PMC article. Review.
Cited by
-
Advantages and Prospects of Tag/Catcher Mediated Antigen Display on Capsid-Like Particle-Based Vaccines.Viruses. 2020 Feb 6;12(2):185. doi: 10.3390/v12020185. Viruses. 2020. PMID: 32041299 Free PMC article. Review.
-
A simple strategy for retargeting lentiviral vectors to desired cell types via a disulfide-bond-forming protein-peptide pair.Sci Rep. 2018 Jul 20;8(1):10990. doi: 10.1038/s41598-018-29253-5. Sci Rep. 2018. PMID: 30030466 Free PMC article.
-
New Routes and Opportunities for Modular Construction of Particulate Vaccines: Stick, Click, and Glue.Front Immunol. 2018 Jun 26;9:1432. doi: 10.3389/fimmu.2018.01432. eCollection 2018. Front Immunol. 2018. PMID: 29997617 Free PMC article. Review.
-
A modular and self-adjuvanted multivalent vaccine platform based on porcine circovirus virus-like nanoparticles.J Nanobiotechnology. 2022 Nov 24;20(1):493. doi: 10.1186/s12951-022-01710-4. J Nanobiotechnology. 2022. PMID: 36424615 Free PMC article.
-
Towards Physiologically and Tightly Regulated Vectored Antibody Therapies.Cancers (Basel). 2020 Apr 13;12(4):962. doi: 10.3390/cancers12040962. Cancers (Basel). 2020. PMID: 32295072 Free PMC article. Review.
References
-
- Günzburg WH, Fleuchaus A, Saller R, Salmons B. 1996. Retroviral vector targeting for gene therapy. Cytokines Mol Ther 2:177–184. - PubMed
-
- Fisher A, Stark A. 2017. FDA approval brings first gene therapy to the United States. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm574058.htm
-
- Girard-Gagnepain A, Amirache F, Costa C, Lévy C, Frecha C, Fusil F, Nègre D, Lavillette D, Cosset FL, Verhoeyen E. 2014. Baboon envelope pseudotyped LVs outperform VSV-G-LVs for gene transfer into early-cytokine-stimulated and resting HSCs. Blood 124:1221–1231. doi:10.1182/blood-2014-02-558163. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

