IL-33 is induced in undifferentiated, non-dividing esophageal epithelial cells in eosinophilic esophagitis
- PMID: 29242581
- PMCID: PMC5730585
- DOI: 10.1038/s41598-017-17541-5
IL-33 is induced in undifferentiated, non-dividing esophageal epithelial cells in eosinophilic esophagitis
Abstract
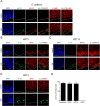
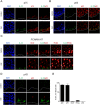
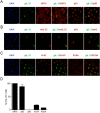
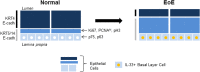
The molecular and cellular etiology of eosinophilic esophagitis (EoE), an emerging tissue-specific allergic disease, involves dysregulated gene expression in esophageal epithelial cells. Herein, we assessed the esophageal expression of IL-33, an epithelium-derived alarmin cytokine, in patients with EoE. IL-33 protein was markedly overexpressed within the nuclei of a subpopulation of basal layer esophageal epithelial cells in patients with active EoE compared to control individuals. IL-33 exhibited dynamic expression as levels normalized upon EoE remission. IL-33-positive basal epithelial cells expressed E-cadherin and the undifferentiated epithelial cell markers keratin 5 and 14 but not the differentiation marker keratin 4. Moreover, the IL-33-positive epithelial cells expressed the epithelial progenitor markers p75 and p63 and lacked the proliferation markers Ki67 and phospho-histone H3. Additionally, the IL-33-positive cells had low expression of PCNA. IL-33 expression was detected in ex vivo-cultured primary esophageal epithelial cells in a subpopulation of cells lacking expression of proliferation markers. Collectively, we report that IL-33 expression is induced in an undifferentiated, non-dividing esophageal epithelial cell population in patients with active EoE.
Conflict of interest statement
M.E.R. is a consultant for N.K.T. Therapeutics, Pulm One, Spoon Guru, Celgene, Shire, Astra Zeneca, and Novartis and has an equity interest in the first three listed and Immune Pharmaceuticals and royalties from reslizumab (Teva Pharmaceuticals). M.E.R. is an inventor of several patents, owned by Cincinnati Children’s. All of the other authors have no potential conflicts to disclose.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

