Infusion dose requirement of rocuronium in patients on phenytoin therapy - A prospective comparative study
- PMID: 29242650
- PMCID: PMC5664883
- DOI: 10.4103/ija.IJA_218_17
Infusion dose requirement of rocuronium in patients on phenytoin therapy - A prospective comparative study
Abstract
Background and aims: Patients with intracranial tumour are usually on anticonvulsants. Patients on phenytoin therapy demonstrate rapid metabolism of nondepolarising muscle relaxants secondary to enzyme induction. Infusion dose requirement of rocuronium in such patients has been sparingly studied. We studied the continuous infusion dose requirement of rocuronium bromide in patients on phenytoin therapy and its correlation with serum levels of phenytoin.
Methods: Seventy-five patients scheduled for supratentorial tumour surgery were included in the study. Patients not on phenytoin were taken as control. The primary outcome variable studied was the infusion dose requirement of rocuronium in patients on phenytoin. Based on pre-operative serum phenytoin levels, study group patients were divided into two groups: sub-therapeutic level group (phenytoin level <10 μg/mL) and therapeutic level group (phenytoin level >10 μg/mL). Following anaesthesia induction, rocuronium bromide 0.6 mg/kg was administered to achieve tracheal intubation. Rocuronium infusion was titrated to maintain zero response on the train-of-four response.
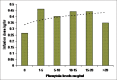
Results: Demographic data were comparable. Patients receiving phenytoin required higher infusion dose compared to the control group (0.429 ± 0.2 mg/kg/h vs. 0.265 ± 0.15 mg/kg/h, P < 0.001). The serum phenytoin level had no correlation to infusion dose requirement of rocuronium (0.429 ± 0.205 mg/kg/h vs. 0.429 ± 0.265 mg/kg/h (P = 0.815). The recovery was faster in the phenytoin group compared to the control group. Haowever, it was not clinically significant.
Conclusion: The infusion dose requirement of rocuronium bromide in patients on phenytoin is higher and the serum levels of phenytoin does not influence the dose required.
Keywords: Infusion; phenytoin; rocuronium.
Conflict of interest statement
There are no conflicts of interest.
Figures
References
-
- Hernández-Palazón J, Tortosa JA, Martínez-Lage JF, Pérez-Ayala M. Rocuronium-induced neuromuscular blockade is affected by chronic phenytoin therapy. J Neurosurg Anesthesiol. 2001;13:79–82. - PubMed
-
- Dahaba AA, Perelman SI, Moskowitz DM, Bennett HL, Shander A, Xiao Z, et al. Geographic regional differences in rocuronium bromide dose-response relation and time course of action: An overlooked factor in determining recommended dosage. Anesthesiology. 2006;104:950–3. - PubMed
-
- Loan PB, Connolly FM, Mirakhur RK, Kumar N, Farling P. Neuromuscular effects of rocuronium in patients receiving beta-adrenoreceptor blocking, calcium entry blocking and anticonvulsant drugs. Br J Anaesth. 1997;78:90–1. - PubMed
-
- Shanks CA, Fragen RJ, Ling D. Continuous intravenous infusion of rocuronium (ORG 9426) in patients receiving balanced, enflurane, or isoflurane anesthesia. Anesthesiology. 1993;78:649–51. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources