The yin and yang of KV channels in cerebral small vessel pathologies
- PMID: 29247493
- PMCID: PMC5760473
- DOI: 10.1111/micc.12436
The yin and yang of KV channels in cerebral small vessel pathologies
Abstract
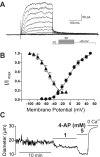
Cerebral SVDs encompass a group of genetic and sporadic pathological processes leading to brain lesions, cognitive decline, and stroke. There is no specific treatment for SVDs, which progress silently for years before becoming clinically symptomatic. Here, we examine parallels in the functional defects of PAs in CADASIL, a monogenic form of SVD, and in response to SAH, a common type of hemorrhagic stroke that also targets the brain microvasculature. Both animal models exhibit dysregulation of the voltage-gated potassium channel, KV 1, in arteriolar myocytes, an impairment that compromises responses to vasoactive stimuli and impacts CBF autoregulation and local dilatory responses to neuronal activity (NVC). However, the extent to which this channelopathy-like defect ultimately contributes to these pathologies is unknown. Combining experimental data with computational modeling, we describe the role of KV 1 channels in the regulation of myocyte membrane potential at rest and during the modest increase in extracellular potassium associated with NVC. We conclude that PA resting membrane potential and myogenic tone depend strongly on KV 1.2/1.5 channel density, and that reciprocal changes in KV channel density in CADASIL and SAH produce opposite effects on extracellular potassium-mediated dilation during NVC.
Keywords: CADASIL; cerebral blood flow; cerebral small vessel disease; subarachnoid hemorrhage; voltage-gated potassium channel.
© 2017 John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



Similar articles
-
Mechanistic insights into a TIMP3-sensitive pathway constitutively engaged in the regulation of cerebral hemodynamics.Elife. 2016 Aug 1;5:e17536. doi: 10.7554/eLife.17536. Elife. 2016. PMID: 27476853 Free PMC article.
-
Potassium channelopathy-like defect underlies early-stage cerebrovascular dysfunction in a genetic model of small vessel disease.Proc Natl Acad Sci U S A. 2015 Feb 17;112(7):E796-805. doi: 10.1073/pnas.1420765112. Epub 2015 Feb 2. Proc Natl Acad Sci U S A. 2015. PMID: 25646445 Free PMC article.
-
Accessory Kvbeta1 subunits differentially modulate the functional expression of voltage-gated K+ channels in mouse ventricular myocytes.Circ Res. 2005 Mar 4;96(4):451-8. doi: 10.1161/01.RES.0000156890.25876.63. Epub 2005 Jan 20. Circ Res. 2005. PMID: 15662035
-
Impact of subarachnoid hemorrhage on parenchymal arteriolar function.Acta Neurochir Suppl. 2013;115:173-7. doi: 10.1007/978-3-7091-1192-5_33. Acta Neurochir Suppl. 2013. PMID: 22890665 Free PMC article. Review.
-
KV channels and the regulation of vascular smooth muscle tone.Microcirculation. 2018 Jan;25(1):10.1111/micc.12421. doi: 10.1111/micc.12421. Microcirculation. 2018. PMID: 28985443 Free PMC article. Review.
Cited by
-
AMPK-α1 or AMPK-α2 Deletion in Smooth Muscles Does Not Affect the Hypoxic Ventilatory Response or Systemic Arterial Blood Pressure Regulation During Hypoxia.Front Physiol. 2018 Jun 6;9:655. doi: 10.3389/fphys.2018.00655. eCollection 2018. Front Physiol. 2018. PMID: 29928235 Free PMC article.
-
PIP2 corrects cerebral blood flow deficits in small vessel disease by rescuing capillary Kir2.1 activity.Proc Natl Acad Sci U S A. 2021 Apr 27;118(17):e2025998118. doi: 10.1073/pnas.2025998118. Proc Natl Acad Sci U S A. 2021. PMID: 33875602 Free PMC article.
-
HB-EGF depolarizes hippocampal arterioles to restore myogenic tone in a genetic model of small vessel disease.Mech Ageing Dev. 2020 Dec;192:111389. doi: 10.1016/j.mad.2020.111389. Epub 2020 Oct 27. Mech Ageing Dev. 2020. PMID: 33127441 Free PMC article.
-
Ex Vivo Pressurized Hippocampal Capillary-Parenchymal Arteriole Preparation for Functional Study.J Vis Exp. 2019 Dec 18;(154):10.3791/60676. doi: 10.3791/60676. J Vis Exp. 2019. PMID: 31904015 Free PMC article.
-
Prostaglandin E2 Dilates Intracerebral Arterioles When Applied to Capillaries: Implications for Small Vessel Diseases.Front Aging Neurosci. 2021 Aug 13;13:695965. doi: 10.3389/fnagi.2021.695965. eCollection 2021. Front Aging Neurosci. 2021. PMID: 34483880 Free PMC article.
References
-
- Yang Z, Lin P-J, Levey A. Monetary costs of dementia in the United States. N Engl J Med. 2013;369(5):489. - PubMed
-
- Pantoni L. Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010;9(7):689–701. - PubMed
-
- Bederson JB, Connolly ES, Batjer HH, Dacey RG, Dion JE, Diringer MN, Duldner JE, Jr, Harbaugh RE, Patel AB, Rosenwasser RH. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a statement for healthcare professionals from a special writing group of the Stroke Council, American Heart Association. Stroke. 2009;40(3):994–1025. - PubMed
-
- Chabriat H, Joutel A, Dichgans M, Tournier-Lasserve E, Bousser M-G. CADASIL. Lancet Neurol. 2009;8(7):643–653. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

