Paying Research Participants: Regulatory Uncertainty, Conceptual Confusion, and a Path Forward
- PMID: 29249912
- PMCID: PMC5728432
Paying Research Participants: Regulatory Uncertainty, Conceptual Confusion, and a Path Forward
Abstract
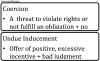
The practice of offering payment to individuals in exchange for their participation in clinical research is widespread and longstanding. Nevertheless, such payment remains the source of substantial debate, in particular about whether or the extent to which offers of payment coerce and/or unduly induce individuals to participate. Yet, the various laws, regulations, and ethical guidelines that govern the conduct of human subjects research offer relatively little in the way of specific guidance regarding what makes a payment offer ethically acceptable-or not. Moreover, there is a lack of definitional agreement regarding what the terms coercion and undue inducement mean in the human subjects research context. It is, therefore, unsurprising that investigators and Institutional Review Boards (IRBs) experience confusion about how to evaluate offers of payment, and lean toward conservative approaches. These trends are exemplified by our pilot data regarding the ways in which some IRB members and investigators (mis)understand the concepts of coercion and undue inducement, as well as the ways in which certain research institutions oversee offers of payment at a local level. This article systematically examines the legal and ethical dimensions of offering payment to research participants. It argues that many concerns about offers of payment to research participants can be attributed to the misguided view that such offers ought to be treated differently than offers of payment in other contexts, a form of "research exceptionalism." We show that rejection of research exceptionalism with respect to payment helps settle open debates about both how best to define coercion and undue influence, and how to understand the relation between these concepts and offers of payment. We argue for adoption of our preferred definitions, ideally by regulatory authorities, and against the conventional conservatism toward payment of research participants. Instead, we draw attention to the rarely asked, even radical, question: are research participants paid enough? We conclude by arguing that we ought to change the default to favor, rather than encourage suspicion of, offers of payment to research participants.
Figures
Similar articles
-
Payment for research participation: a coercive offer?J Med Ethics. 2008 May;34(5):389-92. doi: 10.1136/jme.2007.021857. J Med Ethics. 2008. PMID: 18448723
-
Making the case for completion bonuses in clinical trials.Clin Trials. 2019 Apr;16(2):176-182. doi: 10.1177/1740774518820503. Epub 2018 Dec 20. Clin Trials. 2019. PMID: 30567460
-
Misconceptions about coercion and undue influence: reflections on the views of IRB members.Bioethics. 2013 Nov;27(9):500-7. doi: 10.1111/j.1467-8519.2012.01972.x. Epub 2012 Apr 12. Bioethics. 2013. PMID: 22493972 Free PMC article.
-
Economic vulnerability and payment for research participation.Clin Trials. 2020 Jun;17(3):264-272. doi: 10.1177/1740774520905596. Epub 2020 Feb 17. Clin Trials. 2020. PMID: 32063065 Review.
-
Ending concerns about undue inducement.J Law Med Ethics. 2004 Spring;32(1):100-5. doi: 10.1111/j.1748-720x.2004.tb00453.x. J Law Med Ethics. 2004. PMID: 15152431 Review. No abstract available.
Cited by
-
Identifying Research Priorities to Promote the Well-Being of Family Caregivers of Canadians with Intellectual and/or Developmental Disabilities: A Pilot Delphi Study.Int J Environ Res Public Health. 2023 Nov 16;20(22):7072. doi: 10.3390/ijerph20227072. Int J Environ Res Public Health. 2023. PMID: 37998303 Free PMC article.
-
A Brief Mindfulness-Based Family Psychoeducation Intervention for Chinese Young Adults With First Episode Psychosis: A Study Protocol.Front Psychol. 2019 Mar 11;10:516. doi: 10.3389/fpsyg.2019.00516. eCollection 2019. Front Psychol. 2019. PMID: 30915004 Free PMC article.
-
Impact of COVID-19 on services for people with disabilities and chronic health conditions.Disabil Health J. 2021 Jul;14(3):101090. doi: 10.1016/j.dhjo.2021.101090. Epub 2021 Mar 5. Disabil Health J. 2021. PMID: 33715986 Free PMC article.
-
Exploring patients' adherence to antibiotics by understanding their health knowledge and relational communication in encounters with pharmacists and physicians.Explor Res Clin Soc Pharm. 2023 Nov 22;12:100372. doi: 10.1016/j.rcsop.2023.100372. eCollection 2023 Dec. Explor Res Clin Soc Pharm. 2023. PMID: 38089697 Free PMC article.
-
When money talks: Judging risk and coercion in high-paying clinical trials.PLoS One. 2020 Jan 31;15(1):e0227898. doi: 10.1371/journal.pone.0227898. eCollection 2020. PLoS One. 2020. PMID: 32005037 Free PMC article.
References
-
- Chan Sewell. 6 Hospitalized, One of Them Brain-Dead, After Drug Trial in France. NY Times; Jan 15, 2016. http://www.nytimes.com/2016/01/16/world/europe/french-drug-trial-hospita... [ https://perma.cc/H4LQ-BU73]
-
- Brosky John, Sheridan Cormac. Six Hospitalized in Bial Clinical Trial in France. BioWorld; http://www.bioworld.com/content/six-hospitalized-bial-clinical-trial-france [ https://perma.cc/NM6D-KC2C]
-
- Butler Declan, Callaway Ewen. Scientists in the Dark After French Clinical Trial Proves Fatal. Nature. 2016;529:263. 263. - PubMed
-
- Enserink Martin. More Details Emerge on Fateful French Drug Trial. Science. 2016 Jan 16; http://www.sciencemag.org/news/2016/01/more-details-emerge-fateful-frenc... [ https://perma.cc/6HFB-TTNL]
-
- Butler Declan, Callaway Ewen. Researchers Question Design of Fatal French Clinical Trial. Nature. 2016 Jan 22; http://www.nature.com/news/researchers-question-design-of-fatal-french-c... [ https://perma.cc/J5JG-6JLJ] - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical