Synthesis and biological evaluation of 2-styrylquinolines as antitumour agents and EGFR kinase inhibitors: molecular docking study
- PMID: 29251017
- PMCID: PMC7012010
- DOI: 10.1080/14756366.2017.1407926
Synthesis and biological evaluation of 2-styrylquinolines as antitumour agents and EGFR kinase inhibitors: molecular docking study
Abstract
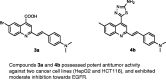
A new series of 4,6-disubstituted 2-(4-(dimethylamino)styryl)quinoline 4a,b-9a,b was synthesized by the reaction of 2-(4-(dimethylamino)styryl)-6-substituted quinoline-4-carboxylic acids 3a,b with thiosemicarbazide, p-hydroxybenzaldehyde, ethylcyanoacetate, and 2,4-pentandione. In addition, the antitumour activity of all synthesized compounds 3a,b-9a,b was studied via MTT assay against two cancer cell lines (HepG2 and HCT116). Furthermore, epidermal growth factor receptor (EGFR) inhibition, using the most potent antitumour compounds, 3a, 3b, 4a, 4b, and 8a, was evaluated. The interpretation of the results showed clearly that the derivatives 3a, 4a, and 4b exhibited the highest antitumour activities against the tested cell lines HepG2 and HCT116 with IC50 range of 7.7-14.2 µg/ml, in comparison with the reference drugs 5-fluorouracil (IC50 = 7.9 and 5.3 µg/ml, respectively) and afatinib (IC50 = 5.4 and 11.4 µg/ml, respectively). In vitro EGFR screening showed that compounds 3a, 3b, 4a, 4b, and 8a exhibited moderate inhibition towards EGFR with IC50 values at micromolar levels (IC50 range of 16.01-1.11 µM) compared with the reference drugs sorafenib (IC50 = 1.14 µM) and erlotinib (IC50 = 0.1 µM). Molecular docking was performed to study the mode of interaction of compounds 3a and 4b with EGFR kinase.
Keywords: EGFR kinase inhibitors; Styrylquinoline; antitumour; molecular docking; synthesis; thiadiazole.
Figures
Similar articles
-
Synthesis, antitumour and antioxidant activities of novel α,β-unsaturated ketones and related heterocyclic analogues: EGFR inhibition and molecular modelling study.J Enzyme Inhib Med Chem. 2018 Dec;33(1):507-518. doi: 10.1080/14756366.2018.1434519. J Enzyme Inhib Med Chem. 2018. PMID: 29455554 Free PMC article.
-
Design, synthesis and biological evaluation of new series of hexahydroquinoline and fused quinoline derivatives as potent inhibitors of wild-type EGFR and mutant EGFR (L858R and T790M).Bioorg Chem. 2020 Dec;105:104274. doi: 10.1016/j.bioorg.2020.104274. Epub 2020 Sep 12. Bioorg Chem. 2020. PMID: 33339080
-
Synthesis and in vitro anticancer evaluation of some fused indazoles, quinazolines and quinolines as potential EGFR inhibitors.Bioorg Chem. 2019 Aug;89:102985. doi: 10.1016/j.bioorg.2019.102985. Epub 2019 May 15. Bioorg Chem. 2019. PMID: 31121559
-
Novel benzothiazole-based dual VEGFR-2/EGFR inhibitors targeting breast and liver cancers: Synthesis, cytotoxic activity, QSAR and molecular docking studies.Bioorg Med Chem Lett. 2022 Feb 15;58:128529. doi: 10.1016/j.bmcl.2022.128529. Epub 2022 Jan 7. Bioorg Med Chem Lett. 2022. PMID: 35007724 Review.
-
2-Anilinopyrimidine derivatives: Design, synthesis, in vitro anti-proliferative activity, EGFR and ARO inhibitory activity, cell cycle analysis and molecular docking study.Bioorg Chem. 2020 Jun;99:103798. doi: 10.1016/j.bioorg.2020.103798. Epub 2020 Mar 29. Bioorg Chem. 2020. PMID: 32247112 Review.
Cited by
-
Acylacetylenes in multiple functionalization of hydroxyquinolines and quinolones.Tetrahedron. 2020 Oct 23;76(43):131523. doi: 10.1016/j.tet.2020.131523. Epub 2020 Aug 20. Tetrahedron. 2020. PMID: 32839627 Free PMC article.
-
Design, synthesis, and carbonic anhydrase inhibition activities of Schiff bases incorporating benzenesulfonamide scaffold: Molecular docking application.Saudi Pharm J. 2023 Dec;31(12):101866. doi: 10.1016/j.jsps.2023.101866. Epub 2023 Nov 10. Saudi Pharm J. 2023. PMID: 38033749 Free PMC article.
-
First synthesis of novel 2,4-bis((E)-styryl)quinoline-3-carboxylate derivatives and their antitumor activity.RSC Adv. 2018 Nov 19;8(68):38844-38849. doi: 10.1039/c8ra08023b. eCollection 2018 Nov 16. RSC Adv. 2018. PMID: 35558278 Free PMC article.
-
Uracil as a Zn-Binding Bioisostere of the Allergic Benzenesulfonamide in the Design of Quinoline-Uracil Hybrids as Anticancer Carbonic Anhydrase Inhibitors.Pharmaceuticals (Basel). 2022 Apr 19;15(5):494. doi: 10.3390/ph15050494. Pharmaceuticals (Basel). 2022. PMID: 35631321 Free PMC article.
-
Remarkable utilization of quinazoline-based homosulfonamide for in vitro cytotoxic effects with triple kinase inhibition activities: cell cycle analysis and molecular docking profile.RSC Adv. 2025 Jan 6;15(1):541-558. doi: 10.1039/d4ra07174c. eCollection 2025 Jan 2. RSC Adv. 2025. PMID: 39763619 Free PMC article.
References
-
- Alanazi AM, Abdel-Aziz AA-M, Shawer TZ, et al. . Synthesis, antitumor and antimicrobial activity of some new 6-methyl-3-phenyl-4(3H)-quinazolinone analogues: in silico studies. J Enzyme Inhib Med Chem 2016;31:721–35. - PubMed
-
- Mohamed MA, Ayyad RR, Shawer TZ, et al. . Synthesis and antitumor evaluation of trimethoxyanilides based on 4(3H)-quinazolinone scaffolds. Eur J Med Chem 2016;112:106–13. - PubMed
-
- Abdel-Aziz AA-M, El-Azab AS, El-Subbagh HI, et al. . Design, synthesis, single-crystal and preliminary antitumor activity of novel arenesulfonylimidazolidin-2-ones. Bioorg Med Chem Lett 2012;22:2008–14. - PubMed
-
- Avendańo C, Menéndez J.. Medicinal chemistry of anticancer agents. Amsterdam: Elsevier; 2008.
-
- Varmus H. The new era in cancer research. Science 2006;312:1162–5. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
