Synthesis and Biocompatibility Studies of New Iminodiacetic Acid Derivatives
- PMID: 29258275
- PMCID: PMC6149924
- DOI: 10.3390/molecules22122265
Synthesis and Biocompatibility Studies of New Iminodiacetic Acid Derivatives
Abstract
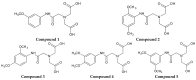
Background: Iminodiacetic acid (IDA) derivatives can be used as ligands to form complexes with technetium, with potential application as hepatobiliary diagnostic agents. The aim of this study was to synthesize five novel IDA derivatives and to compare their effects on plasma haemostasis with clinically approved ligands for technetium complexation.
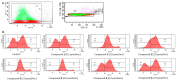
Methods: The influence of synthesized IDA derivatives on plasma haemostasis was evaluated spectrophotometrically by clot formation and lysis test (CL-test), coagulation assay, Prothrombin Time and Activated Partial Tromboplastin Time. The effects of the tested compounds on erythrocytes were assessed using haemolysis assays, microscopy and flow cytometry studies.
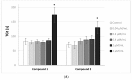
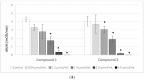
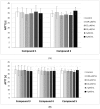
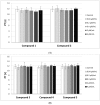
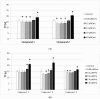
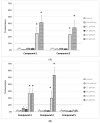
Results: Despite their significant influence on the kinetic parameters of the process of clot formation and fibrinolysis, the tested ligands, at potential diagnostic concentrations, did not alter the overall potential of clot formation and lysis (CLAUC). At potential diagnostic concentrations (0.4 μmol/mL) all the tested compounds showed no adverse effects on the membranes of RBCs (Red Blood Cells).
Conclusion: IDA derivatives with methoxy substituents in aromatic ring, exert multidirectional effects on plasma haemostasis and should be considered safe as their significant impacts were mostly observed at 4 μmol/mL, which is about 10-fold higher than the theoretical plasma concentrations of these compounds.
Keywords: biocompatibility; haemostasis; iminodiacetic acid; radiopharmaceuticals.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Biocompatibility Studies of Gadolinium Complexes with Iminodiacetic Acid Derivatives.Biol Trace Elem Res. 2019 Jun;189(2):426-436. doi: 10.1007/s12011-018-1496-6. Epub 2018 Sep 13. Biol Trace Elem Res. 2019. PMID: 30215189 Free PMC article.
-
Stability of erythrocyte membrane and overall hemostasis potential - A biocompatibility study of mebrofenin and other iminodiacetic acid derivatives.Pharmacol Rep. 2015 Dec;67(6):1230-9. doi: 10.1016/j.pharep.2015.05.021. Epub 2015 Jun 4. Pharmacol Rep. 2015. PMID: 26481547
-
Biocompatible sulfenamide and sulfonamide derivatives of metformin can exert beneficial effects on plasma haemostasis.Chem Biol Interact. 2018 Jan 25;280:15-27. doi: 10.1016/j.cbi.2017.12.005. Epub 2017 Dec 5. Chem Biol Interact. 2018. PMID: 29217384
-
New prodrugs of metformin do not influence the overall haemostasis potential and integrity of the erythrocyte membrane.Eur J Pharmacol. 2017 Sep 15;811:208-221. doi: 10.1016/j.ejphar.2017.06.011. Epub 2017 Jun 10. Eur J Pharmacol. 2017. PMID: 28606852
-
Technetium-99m-iminodiacetic acid organic anions: review of biokinetics and clinical application in hepatology.Hepatology. 1989 Jan;9(1):139-53. doi: 10.1002/hep.1840090123. Hepatology. 1989. PMID: 2642291 Review.
Cited by
-
Biocompatibility Studies of Gadolinium Complexes with Iminodiacetic Acid Derivatives.Biol Trace Elem Res. 2019 Jun;189(2):426-436. doi: 10.1007/s12011-018-1496-6. Epub 2018 Sep 13. Biol Trace Elem Res. 2019. PMID: 30215189 Free PMC article.
References
-
- Świeczko-Żurek B. Biomateriały (Biomaterials) Wydawnictwo Politechniki Gdańskiej; Gdańsk, Poland: 2009. Biomedical Engineering.
-
- U.S. Department of Health and Human Services. Food and Drug Administration. Center for Devices and Radiological Health . Use of International Standard ISO 10993-1, Biological Evaluation of Medical Devices—Part 1: Evaluation and Testing within a Risk Management Process. U.S. Department of Health and Human Services; Food and Drug Administration ; Center for Devices and Radiological Health; Washington, DC, USA: 2016. Guidance for Industry and Food and Drug Administration Staff.
-
- Pereira B.T., Ferreira Silva E., Gonçalves M.A., Mancini D.T., Ramalho T.C. Exploring EPR Parameters of 99Tc Complexes for Designing New MRI Probes: Coordination Environment, Solvent, and Thermal Effects on the Spectroscopic Properties. J. Chem. 2017;2017 doi: 10.1155/2017/8102812. - DOI
-
- Kiritsis C., Mavroidi B., Shegani A., Palamaris L., Loudos G., Sagnou M., Pirmettis J., Papadopoulos M., Pelecanou M. 2-(4′-Aminophenyl)benzothiazole Labeled with 99mTc-Cyclopentadienyl for Imaging β-Amyloid Plaques. ACS Med. Chem. Lett. 2017;8:1089–1092. doi: 10.1021/acsmedchemlett.7b00294. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

