Global inactivation of carboxylesterase 1 (Ces1/Ces1g) protects against atherosclerosis in Ldlr -/- mice
- PMID: 29259301
- PMCID: PMC5736751
- DOI: 10.1038/s41598-017-18232-x
Global inactivation of carboxylesterase 1 (Ces1/Ces1g) protects against atherosclerosis in Ldlr -/- mice
Abstract
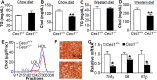
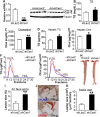
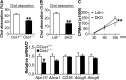
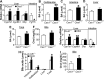
Atherosclerotic cardiovascular disease is a leading cause of death in the western world. Increased plasma triglyceride and cholesterol levels are major risk factors for this disease. Carboxylesterase 1 (Ces1/Ces1g) has been shown to play a role in metabolic control. So far, the role of mouse Ces1/Ces1g deficiency in atherosclerosis is not elucidated. We generated Ces1/Ces1g -/- mice. Compared to wild-type mice, Ces1/Ces1g -/- mice had reduced plasma cholesterol levels. We then generated Ces1g -/- Ldlr -/- double knockout (DKO) mice, which were fed a Western diet for 16 weeks. Compared to Ldlr -/- mice, DKO mice displayed decreased plasma cholesterol and TG levels and reduced atherosclerotic lesions. Interestingly, knockdown of hepatic Ces1/Ces1g in Apoe -/- mice resulted in hyperlipidemia and exacerbated Western diet-induced atherogenesis. Mechanistically, global inactivation of Ces1/Ces1g inhibited intestinal cholesterol and fat absorption and Niemann-Pick C1 like 1 expression, and increased macrophage cholesterol efflux by inducing ATP-binding cassette subfamily A member 1 (ABCA1) and ABCG1. Ces1/Ces1g ablation also promoted M2 macrophage polarization and induced hepatic cholesterol 7α-hydroxylase and sterol 12α-hydroxylase expression. In conclusion, global loss of Ces1/Ces1g protects against the development of atherosclerosis by inhibiting intestinal cholesterol and triglyceride absorption and promoting macrophage cholesterol efflux.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures



Similar articles
-
Medium-chain triglycerides promote macrophage reverse cholesterol transport and improve atherosclerosis in ApoE-deficient mice fed a high-fat diet.Nutr Res. 2016 Sep;36(9):964-973. doi: 10.1016/j.nutres.2016.06.004. Epub 2016 Jun 4. Nutr Res. 2016. PMID: 27632916
-
Deficiency of ATP-Binding Cassette Transporters A1 and G1 in Endothelial Cells Accelerates Atherosclerosis in Mice.Arterioscler Thromb Vasc Biol. 2016 Jul;36(7):1328-37. doi: 10.1161/ATVBAHA.115.306670. Epub 2016 May 19. Arterioscler Thromb Vasc Biol. 2016. PMID: 27199450 Free PMC article.
-
Macrophage miR-34a Is a Key Regulator of Cholesterol Efflux and Atherosclerosis.Mol Ther. 2020 Jan 8;28(1):202-216. doi: 10.1016/j.ymthe.2019.09.008. Epub 2019 Sep 12. Mol Ther. 2020. PMID: 31604677 Free PMC article.
-
Melanocortin 1 Receptor Deficiency Promotes Atherosclerosis in Apolipoprotein E-/- Mice.Arterioscler Thromb Vasc Biol. 2018 Feb;38(2):313-323. doi: 10.1161/ATVBAHA.117.310418. Epub 2017 Dec 28. Arterioscler Thromb Vasc Biol. 2018. PMID: 29284608 Free PMC article.
-
Vitamin D Protects Against Atherosclerosis via Regulation of Cholesterol Efflux and Macrophage Polarization in Hypercholesterolemic Swine.Arterioscler Thromb Vasc Biol. 2015 Nov;35(11):2432-42. doi: 10.1161/ATVBAHA.115.306132. Epub 2015 Sep 17. Arterioscler Thromb Vasc Biol. 2015. PMID: 26381871 Free PMC article.
Cited by
-
The Effects of Diosgenin on Hypolipidemia and Its Underlying Mechanism: A Review.Diabetes Metab Syndr Obes. 2021 Sep 15;14:4015-4030. doi: 10.2147/DMSO.S326054. eCollection 2021. Diabetes Metab Syndr Obes. 2021. PMID: 34552341 Free PMC article. Review.
-
Prenatal inflammation causes obesity and abnormal lipid metabolism via impaired energy expenditure in male offspring.Nutr Metab (Lond). 2022 Feb 8;19(1):8. doi: 10.1186/s12986-022-00642-y. Nutr Metab (Lond). 2022. PMID: 35135573 Free PMC article.
-
Genome-wide study on 72,298 individuals in Korean biobank data for 76 traits.Cell Genom. 2022 Oct 5;2(10):100189. doi: 10.1016/j.xgen.2022.100189. eCollection 2022 Oct 12. Cell Genom. 2022. PMID: 36777999 Free PMC article.
-
Carboxylesterase 1-mediated endocannabinoid metabolism in skin: role in melanoma progression in BRafV600E/Pten-/- mice.Cancer Metab. 2025 Feb 11;13(1):8. doi: 10.1186/s40170-025-00378-2. Cancer Metab. 2025. PMID: 39934865 Free PMC article.
-
Spatially Resolved Activity-based Proteomic Profiles of the Murine Small Intestinal Lipases.Mol Cell Proteomics. 2020 Dec;19(12):2104-2115. doi: 10.1074/mcp.RA120.002171. Epub 2020 Oct 6. Mol Cell Proteomics. 2020. PMID: 33023980 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

