Anti-LINGO-1 has no detectable immunomodulatory effects in preclinical and phase 1 studies
- PMID: 29259995
- PMCID: PMC5732005
- DOI: 10.1212/NXI.0000000000000417
Anti-LINGO-1 has no detectable immunomodulatory effects in preclinical and phase 1 studies
Abstract
Objective: To evaluate whether the anti-LINGO-1 antibody has immunomodulatory effects.
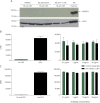
Methods: Human peripheral blood mononuclear cells (hPBMCs), rat splenocytes, and rat CD4+ T cells were assessed to determine whether LINGO-1 was expressed and was inducible. Anti-LINGO-1 Li81 (0.1-30 μg/mL) effect on proliferation/cytokine production was assessed in purified rat CD4+ T cells and hPBMCs stimulated with antibodies to CD3 +/- CD28. In humans, the effect of 2 opicinumab (anti-LINGO-1/BIIB033; 30, 60, and 100 mg/kg) or placebo IV administrations was evaluated in RNA from blood and CSF samples taken before and after administration in phase 1 clinical trials; paired samples were assessed for differentially expressed genes by microarray. RNA from human CSF cell pellets was analyzed by quantitative real-time PCR for changes in transcripts representative of cell types, activation markers, and soluble proteins of the adaptive/innate immune systems. ELISA quantitated the levels of CXCL13 protein in human CSF supernatants.
Results: LINGO-1 is not expressed in hPBMCs, rat splenocytes, or rat CD4+ T cells; LINGO-1 blockade with Li81 did not affect T-cell proliferation or cytokine production from purified rat CD4+ T cells or hPBMCs. LINGO-1 blockade with opicinumab resulted in neither significant changes in immune system gene expression in blood and CSF, nor changes in CXCL13 CSF protein levels (clinical studies).
Conclusions: These data support the hypothesis that LINGO-1 blockade does not affect immune function.
Classification of evidence: This study provides Class II evidence that in patients with MS, opicinumab does not have immunomodulatory effects detected by changes in immune gene transcript expression.
Figures



References
-
- Harrison DM. Multiple sclerosis. Ann Intern Med 2014;160:ITC4-2–ITC4-18. - PubMed
-
- Cadavid D, Balcer L, Galetta S, et al. ; RENEW Study Investigators. Safety and efficacy of opicinumab in acute optic neuritis (RENEW): a randomised, placebo-controlled, phase 2 trial. Lancet Neurol 2017;16:189–199. - PubMed
-
- Tran JQ, Rana J, Barkhof F, et al. Randomized phase I trials of the safety/tolerability of anti-LINGO-1 monoclonal antibody BIIB033. Neurol Neuroimmunol Neuroinflamm 2014;1:e18. doi: 10.1212/NXI.0000000000000018. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
