Clinical translation of handheld optical coherence tomography: practical considerations and recent advancements
- PMID: 29260539
- PMCID: PMC5735247
- DOI: 10.1117/1.JBO.22.12.121715
Clinical translation of handheld optical coherence tomography: practical considerations and recent advancements
Abstract
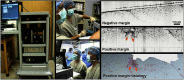
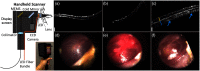
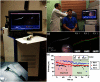
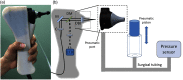
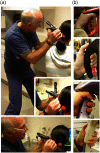
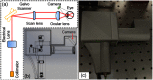
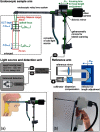
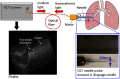
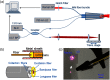
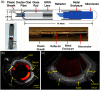
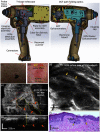
Since the inception of optical coherence tomography (OCT), advancements in imaging system design and handheld probes have allowed for numerous advancements in disease diagnostics and characterization of the structural and optical properties of tissue. OCT system developers continue to reduce form factor and cost, while improving imaging performance (speed, resolution, etc.) and flexibility for applicability in a broad range of fields, and nearly every clinical specialty. An extensive array of components to construct customized systems has also become available, with a range of commercial entities that produce high-quality products, from single components to full systems, for clinical and research use. Many advancements in the development of these miniaturized and portable systems can be linked back to a specific challenge in academic research, or a clinical need in medicine or surgery. Handheld OCT systems are discussed and explored for various applications. Handheld systems are discussed in terms of their relative level of portability and form factor, with mention of the supporting technologies and surrounding ecosystem that bolstered their development. Additional insight from our efforts to implement systems in several clinical environments is provided. The trend toward well-designed, efficient, and compact handheld systems paves the way for more widespread adoption of OCT into point-of-care or point-of-procedure applications in both clinical and commercial settings.
Keywords: diagnostics; handheld; multimodal; optical coherence tomography; point-of-care; system development.
(2017) COPYRIGHT Society of Photo-Optical Instrumentation Engineers (SPIE).
Figures









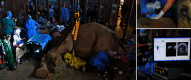







References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

