Peptide aptamer-modified single-walled carbon nanotube-based transistors for high-performance biosensors
- PMID: 29263412
- PMCID: PMC5738443
- DOI: 10.1038/s41598-017-18169-1
Peptide aptamer-modified single-walled carbon nanotube-based transistors for high-performance biosensors
Abstract
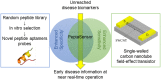
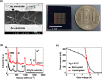
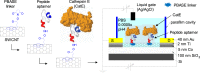
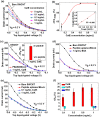
Biosensors employing single-walled carbon nanotube field-effect transistors (SWCNT FETs) offer ultimate sensitivity. However, besides the sensitivity, a high selectivity is critically important to distinguish the true signal from interference signals in a non-controlled environment. This work presents the first demonstration of the successful integration of a novel peptide aptamer with a liquid-gated SWCNT FET to achieve highly sensitive and specific detection of Cathepsin E (CatE), a useful prognostic biomarker for cancer diagnosis. Novel peptide aptamers that specifically recognize CatE are engineered by systemic in vitro evolution. The SWCNTs were firstly grown using the thermal chemical vapor deposition (CVD) method and then were employed as a channel to fabricate a SWCNT FET device. Next, the SWCNTs were functionalized by noncovalent immobilization of the peptide aptamer using 1-pyrenebutanoic acid succinimidyl ester (PBASE) linker. The resulting FET sensors exhibited a high selectivity (no response to bovine serum albumin and cathepsin K) and label-free detection of CatE at unprecedentedly low concentrations in both phosphate-buffered saline (2.3 pM) and human serum (0.23 nM). Our results highlight the use of peptide aptamer-modified SWCNT FET sensors as a promising platform for near-patient testing and point-of-care testing applications.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
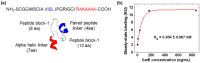

Similar articles
-
Ultrasensitive Label-Free Sensing of IL-6 Based on PASE Functionalized Carbon Nanotube Micro-Arrays with RNA-Aptamers as Molecular Recognition Elements.Biosensors (Basel). 2017 Apr 17;7(2):17. doi: 10.3390/bios7020017. Biosensors (Basel). 2017. PMID: 28420169 Free PMC article.
-
Label-free protein biosensor based on aptamer-modified carbon nanotube field-effect transistors.Anal Chem. 2007 Jan 15;79(2):782-7. doi: 10.1021/ac060830g. Anal Chem. 2007. PMID: 17222052
-
Diagnostics of Tuberculosis with Single-Walled Carbon Nanotube-Based Field-Effect Transistors.ACS Sens. 2024 Apr 26;9(4):1957-1966. doi: 10.1021/acssensors.3c02694. Epub 2024 Mar 14. ACS Sens. 2024. PMID: 38484361 Free PMC article.
-
Toward the Commercialization of Carbon Nanotube Field Effect Transistor Biosensors.Biosensors (Basel). 2023 Feb 27;13(3):326. doi: 10.3390/bios13030326. Biosensors (Basel). 2023. PMID: 36979538 Free PMC article. Review.
-
Carbon Nanotube-Based Field-Effect Transistor Biosensors for Biomedical Applications: Decadal Developments and Advancements (2016-2025).Biosensors (Basel). 2025 May 7;15(5):296. doi: 10.3390/bios15050296. Biosensors (Basel). 2025. PMID: 40422035 Free PMC article. Review.
Cited by
-
Peptide-Functionalized Carbon Nanotube Chemiresistors: The Effect of Nanotube Density on Gas Sensing.Sensors (Basel). 2023 Oct 14;23(20):8469. doi: 10.3390/s23208469. Sensors (Basel). 2023. PMID: 37896562 Free PMC article.
-
Applications of Adductomics in Chemically Induced Adverse Outcomes and Major Emphasis on DNA Adductomics: A Pathbreaking Tool in Biomedical Research.Int J Mol Sci. 2021 Sep 20;22(18):10141. doi: 10.3390/ijms221810141. Int J Mol Sci. 2021. PMID: 34576304 Free PMC article. Review.
-
Fabrication and Functionalisation of Nanocarbon-Based Field-Effect Transistor Biosensors.Chembiochem. 2022 Dec 5;23(23):e202200282. doi: 10.1002/cbic.202200282. Epub 2022 Nov 3. Chembiochem. 2022. PMID: 36193790 Free PMC article. Review.
-
Recent Advancements in Aptamer-Based Surface Plasmon Resonance Biosensing Strategies.Biosensors (Basel). 2021 Jul 10;11(7):233. doi: 10.3390/bios11070233. Biosensors (Basel). 2021. PMID: 34356703 Free PMC article. Review.
-
An Ultrasensitive Norfentanyl Sensor Based on a Carbon Nanotube-Based Field-Effect Transistor for the Detection of Fentanyl Exposure.ACS Appl Mater Interfaces. 2023 Aug 9;15(31):37784-37793. doi: 10.1021/acsami.3c05958. Epub 2023 Jul 31. ACS Appl Mater Interfaces. 2023. PMID: 37523478 Free PMC article.
References
-
- Liu S, Guo X. Carbon nanomaterials field-effect-transistor-based biosensors. NPG Asia Materials. 2012;4:e23. doi: 10.1038/am.2012.42. - DOI
-
- Maehashi K, Matsumoto K, Kerman K, Takamura Y, Tamiya E. Ultrasensitive Detection of DNA Hybridization Using Carbon Nanotube Field-Effect Transistors. Jpn. J. Appl. Phys. 2004;43:L1558–L1560. doi: 10.1143/JJAP.43.L1558. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

