Drug transport mechanisms and in vitro release kinetics of vancomycin encapsulated chitosan-alginate polyelectrolyte microparticles as a controlled drug delivery system
- PMID: 29269322
- PMCID: PMC5805659
- DOI: 10.1016/j.ejps.2017.12.012
Drug transport mechanisms and in vitro release kinetics of vancomycin encapsulated chitosan-alginate polyelectrolyte microparticles as a controlled drug delivery system
Abstract
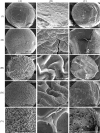
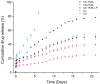
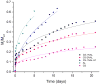
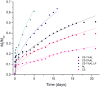
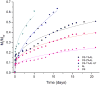
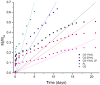
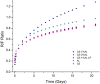
In this study, chitosan-alginate polyelectrolyte microparticles containing the antibiotic, vancomycin chloride were prepared using the ionotropic gelation (coacervation) technique. In vitro release and drug transport mechanisms were studied concerning the chitosan only and alginate only microparticles as a control group. Further, the effect of porosity on the drug transport mechanism was also studied for chitosan-alginate mixed particles produced by lyophilizing in contrast to the air-dried non-porous particles. According to the in vitro release data, alginate only and chitosan only microparticles showed burst release and prolonged release respectively. Chitosan-alginate lyophilized microparticles showed the best-controlled release of vancomycin with the average release of 22μg per day for 14days. Also, when increasing alginate concentration there was no increase in the release rate of vancomycin. The release data of all the microparticles were treated with Ritger-Peppas, Higuchi, Peppas-Sahlin, zero-order, and first-order kinetic models. The best fit was observed with Peppas-Sahlin model, indicating the drug transport mechanism was controlled by both Fickian diffusion and case II relaxations. Also, Fickian diffusion dominates the drug transport mechanism of all air-dried samples during the study period. However, the Fickian contribution was gradually reducing with time. Porosity significantly effects the drug transport mechanism as case II relaxation dominates after day 10 of the lyophilized microparticles.
Keywords: Alginate; Chitosan; Lyophilizing; Polyelectrolyte; Transport mechanism.
Copyright © 2017 Elsevier B.V. All rights reserved.
Figures




Similar articles
-
Chitosan-alginate multilayer beads for controlled release of ampicillin.Int J Pharm. 2005 Feb 16;290(1-2):45-54. doi: 10.1016/j.ijpharm.2004.11.015. Epub 2005 Jan 5. Int J Pharm. 2005. PMID: 15664129
-
Polyelectrolyte-coated alginate microspheres as drug delivery carriers for dexamethasone release.Drug Deliv. 2009 Aug;16(6):331-40. doi: 10.1080/10717540903031126. Drug Deliv. 2009. PMID: 19606947 Free PMC article.
-
Chitosan oligosaccharide as prospective cross-linking agent for naproxen-loaded Ca-alginate microparticles with improved pH sensitivity.Drug Dev Ind Pharm. 2013 Jan;39(1):77-88. doi: 10.3109/03639045.2012.658813. Epub 2012 Feb 18. Drug Dev Ind Pharm. 2013. PMID: 22339172
-
Development and evaluation of pH-sensitive sodium alginate/chitosan microparticles containing the antituberculosis drug rifampicin.Mater Sci Eng C Mater Biol Appl. 2014 Jun 1;39:161-7. doi: 10.1016/j.msec.2014.01.054. Epub 2014 Feb 10. Mater Sci Eng C Mater Biol Appl. 2014. PMID: 24863212 Review.
-
Alginate drug delivery systems: application in context of pharmaceutical and biomedical research.Drug Dev Ind Pharm. 2014 Dec;40(12):1576-84. doi: 10.3109/03639045.2014.917657. Epub 2014 Aug 11. Drug Dev Ind Pharm. 2014. PMID: 25109399 Review.
Cited by
-
Kinetics, Equilibrium, and Thermodynamics for Conjugation of Chitosan with Insulin-Mimetic [meso-Tetrakis(4-sulfonatophenyl)porphyrinato]oxovanadate(IV)(4-) in an Aqueous Solution.ACS Omega. 2023 Oct 24;8(44):41612-41623. doi: 10.1021/acsomega.3c05804. eCollection 2023 Nov 7. ACS Omega. 2023. PMID: 37970023 Free PMC article.
-
Poly(Lactic Acid)-Based Microparticles for Drug Delivery Applications: An Overview of Recent Advances.Pharmaceutics. 2022 Feb 4;14(2):359. doi: 10.3390/pharmaceutics14020359. Pharmaceutics. 2022. PMID: 35214091 Free PMC article. Review.
-
Formulation of Piperine Nanoparticles: In Vitro Breast Cancer Cell Line and In Vivo Evaluation.Polymers (Basel). 2022 Mar 26;14(7):1349. doi: 10.3390/polym14071349. Polymers (Basel). 2022. PMID: 35406223 Free PMC article.
-
The Oral Administration of Lidocaine HCl Biodegradable Microspheres: Formulation and Optimization.Int J Nanomedicine. 2020 Feb 5;15:857-869. doi: 10.2147/IJN.S236273. eCollection 2020. Int J Nanomedicine. 2020. PMID: 32103942 Free PMC article.
-
Natural-Based Biomaterial for Skin Wound Healing (Gelatin vs. Collagen): Expert Review.Polymers (Basel). 2021 Jul 14;13(14):2319. doi: 10.3390/polym13142319. Polymers (Basel). 2021. PMID: 34301076 Free PMC article. Review.
References
-
- Alvarez-Lorenzo C, Concheiro A, Dubovik AS, Grinberg NV, Burova TV, Grinberg VY. Temperature-sensitive chitosan-poly(N-isopropylacrylamide) interpenetrated networks with enhanced loading capacity and controlled release properties. J Control Release. 2005;102:629–641. doi: 10.1016/j.jconrel.2004.10.021. - DOI - PubMed
-
- Borges O, Cordeiro-da-Silva A, Romeijn SG, Amidi M, de Sousa A, Borchard G, Junginger HE. Uptake studies in rat Peyer’s patches, cytotoxicity and release studies of alginate coated chitosan nanoparticles for mucosal vaccination. J Control Release. 2006;114:348–358. doi: 10.1016/j.jconrel.2006.06.011. - DOI - PubMed
-
- Buzia OD, DIMA C, DIMA S. Preparation and characterization of chitosan microspheres for vancomycin delivery. Parmacia. 2015;63:6.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

