Pollen-Expressed Leucine-Rich Repeat Extensins Are Essential for Pollen Germination and Growth
- PMID: 29269573
- PMCID: PMC5841703
- DOI: 10.1104/pp.17.01241
Pollen-Expressed Leucine-Rich Repeat Extensins Are Essential for Pollen Germination and Growth
Abstract
During pollen tube growth, the walls of the tube provide the mechanical strength resisting turgor pressure to protect two sperm cells. Cell wall proteins may play an important role in this process. Pollen tube cell wall proteins known as leucine-rich repeat extensins (LRXs) harbor a leucine-rich repeat domain and an extensin domain. In this study, the functions of four pollen-expressed LRXs, LRX8, LRX9, LRX10, and LRX11 (LRX8-11), were characterized in Arabidopsis (Arabidopsis thaliana). LRX8-11 displayed a consistent expression pattern in mature pollen grains and pollen tubes. In a phenotypic analysis of four single mutants, six double mutants, four triple mutants, and a quadruple mutant, the triple and quadruple mutant plants displayed markedly reduced seed set and decreased male transmission efficiency accompanied by compromised pollen germination and pollen tube growth. GFP-fused LRX8, LRX10, and LRX11 were found to be localized to pollen tube cell walls. An immunohistochemical analysis of pollen tube cell wall polysaccharides showed an increase in the amount of rhamnogalacturonan I in the subapical walls of pollen tubes of the lrx9 lrx10 lrx11 and lrx8 lrx9 lrx11 mutants and a decrease in the content of fucosylated xyloglucans in lrx8 lrx9 lrx11 compared with wild-type plants. Moreover, the callose content in the apical walls of pollen tubes increased in the lrx8 lrx9 lrx11 mutant. In conclusion, we propose that LRX8-11 function synergistically to maintain pollen tube cell wall integrity; thus, they play critical roles in pollen germination and pollen tube growth.
© 2018 American Society of Plant Biologists. All Rights Reserved.
Figures
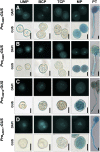


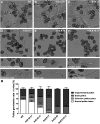


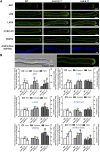
Comment in
-
Coordinating Cell Walls and Cell Growth: A Role for LRX Extensin Chimeras.Plant Physiol. 2018 Mar;176(3):1890-1891. doi: 10.1104/pp.18.00084. Plant Physiol. 2018. PMID: 29630498 Free PMC article. No abstract available.
References
-
- Alexander MP. (1969) Differential staining of aborted and nonaborted pollen. Stain Technol 44: 117–122 - PubMed
-
- Baron-Epel O, Gharyal PK, Schindler M (1988) Pectins as mediators of wall porosity in soybean cells. Planta 175: 389–395 - PubMed
-
- Baumberger N, Doesseger B, Guyot R, Diet A, Parsons RL, Clark MA, Simmons MP, Bedinger P, Goff SA, Ringli C, et al. (2003a) Whole-genome comparison of leucine-rich repeat extensins in Arabidopsis and rice: a conserved family of cell wall proteins form a vegetative and a reproductive clade. Plant Physiol 131: 1313–1326 - PMC - PubMed
-
- Baumberger N, Steiner M, Ryser U, Keller B, Ringli C (2003b) Synergistic interaction of the two paralogous Arabidopsis genes LRX1 and LRX2 in cell wall formation during root hair development. Plant J 35: 71–81 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

