Chronic limb-threatening ischemia could benefit from growth hormone therapy for wound healing and limb salvage
- PMID: 29271292
- PMCID: PMC5772430
- DOI: 10.1177/1753944717745494
Chronic limb-threatening ischemia could benefit from growth hormone therapy for wound healing and limb salvage
Abstract
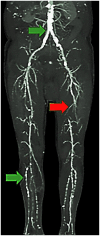
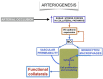
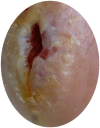
Revascularization for chronic limb-threatening ischemia (CLTI) is necessary to alleviate symptoms and wound healing. When it fails or is not possible, there are few alternatives to avoid limb amputation in these patients. Although experimental studies with stem cells and growth factors have shown promise, clinical trials have demonstrated inconsistent results because CLTI patients generally need arteriogenesis rather than angiogenesis. Moreover, in addition to the perfusion of the limb, there is the need to improve the neuropathic response for wound healing, especially in diabetic patients. Growth hormone (GH) is a pleiotropic hormone capable of boosting the aforementioned processes and adds special benefits for the redox balance. This hormone has the potential to mitigate symptoms in ischemic patients with no other options and improves the cardiovascular complications associated with the disease. Here, we discuss the pros and cons of using GH in such patients, focus on its effects on peripheral arteries, and analyze the possible benefits of treating CLTI with this hormone.
Keywords: angiogenesis; arteriogenesis; chronic limb-threatening ischemia; growth hormone; neuropathic response; redox balance; wound healing.
Conflict of interest statement
Figures



Similar articles
-
Impact of angiosome- and nonangiosome-targeted peroneal bypass on limb salvage and healing in patients with chronic limb-threatening ischemia.J Vasc Surg. 2017 Nov;66(5):1479-1487. doi: 10.1016/j.jvs.2017.04.074. Epub 2017 Jul 26. J Vasc Surg. 2017. PMID: 28756043
-
Clinical outcomes of bypass-first versus endovascular-first strategy in patients with chronic limb-threatening ischemia due to infrageniculate arterial disease.J Vasc Surg. 2019 Jan;69(1):156-163.e1. doi: 10.1016/j.jvs.2018.05.244. J Vasc Surg. 2019. PMID: 30579443 Free PMC article.
-
PROMISE I: Early feasibility study of the LimFlow System for percutaneous deep vein arterialization in no-option chronic limb-threatening ischemia: 12-month results.J Vasc Surg. 2021 Nov;74(5):1626-1635. doi: 10.1016/j.jvs.2021.04.057. Epub 2021 May 18. J Vasc Surg. 2021. PMID: 34019990 Clinical Trial.
-
Medical adjunctive therapy for patients with chronic limb-threatening ischemia: a systematic review.J Cardiovasc Surg (Torino). 2019 Dec;60(6):642-651. doi: 10.23736/S0021-9509.19.11108-1. Epub 2019 Oct 9. J Cardiovasc Surg (Torino). 2019. PMID: 31603294
-
Technical Aspects of Percutaneous Deep Venous Arterialization Using Off-the-Shelf Devices.J Endovasc Ther. 2024 Jun;31(3):350-359. doi: 10.1177/15266028221127850. Epub 2022 Sep 28. J Endovasc Ther. 2024. PMID: 36168745 Review.
Cited by
-
Growth Hormone (GH) and Cardiovascular System.Int J Mol Sci. 2018 Jan 18;19(1):290. doi: 10.3390/ijms19010290. Int J Mol Sci. 2018. PMID: 29346331 Free PMC article. Review.
-
HUVECs-derived exosomes increase neovascularization and decrease limb necrosis in hindlimb ischemia.Narra J. 2024 Dec;4(3):e1358. doi: 10.52225/narra.v4i3.1358. Epub 2024 Nov 22. Narra J. 2024. PMID: 39816111 Free PMC article.
-
The Role of Midkine in Arteriogenesis, Involving Mechanosensing, Endothelial Cell Proliferation, and Vasodilation.Int J Mol Sci. 2018 Aug 29;19(9):2559. doi: 10.3390/ijms19092559. Int J Mol Sci. 2018. PMID: 30158425 Free PMC article. Review.
-
The Role of Growth Hormone on Ovarian Functioning and Ovarian Angiogenesis.Front Endocrinol (Lausanne). 2019 Jul 16;10:450. doi: 10.3389/fendo.2019.00450. eCollection 2019. Front Endocrinol (Lausanne). 2019. PMID: 31379735 Free PMC article. Review.
-
Why Should Growth Hormone (GH) Be Considered a Promising Therapeutic Agent for Arteriogenesis? Insights from the GHAS Trial.Cells. 2020 Mar 27;9(4):807. doi: 10.3390/cells9040807. Cells. 2020. PMID: 32230747 Free PMC article. Review.
References
-
- Blanes JI, Cairols MA, Marrugat J; ESTIME. Prevalence of peripheral artery disease and its associated risk factors in Spain: the ESTIME Study. Int Angiol 2009; 28: 20–25. - PubMed
-
- Hirsch AT, Criqui MH, Treat-Jacobson D, et al. Peripheral arterial disease detection, awareness, and treatment in primary care. JAMA 2001; 286: 1317–1324. - PubMed
-
- Belch JJF, Topol EJ, Agnelli G, et al. ; Prevention of Atherothrombotic Disease Network. Critical issues in peripheral arterial disease detection and management: a call to action. Arch Intern Med 2003; 163: 884–892. - PubMed
-
- Fowkes G, Gillespie IN. Angioplasty (versus non surgical management) for intermittent claudication. Cochrane Database Syst Rev 2000;(2):CD000017. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

