IGF1 receptor and thyroid-associated ophthalmopathy
- PMID: 29273685
- PMCID: PMC6561656
- DOI: 10.1530/JME-17-0276
IGF1 receptor and thyroid-associated ophthalmopathy
Abstract
Thyroid-associated ophthalmopathy (TAO) is a vexing and poorly understood autoimmune process involving the upper face and tissues surrounding the eyes. In TAO, the orbit can become inflamed and undergo substantial remodeling that is disfiguring and can lead to loss of vision. There are currently no approved medical therapies for TAO, the consequence of its uncertain pathogenic nature. It usually presents as a component of the syndrome known as Graves' disease where loss of immune tolerance to the thyrotropin receptor (TSHR) results in the generation of activating antibodies against that protein and hyperthyroidism. The role for TSHR and these antibodies in the development of TAO is considerably less well established. We have reported over the past 2 decades evidence that the insulin-like growth factorI receptor (IGF1R) may also participate in the pathogenesis of TAO. Activating antibodies against IGF1R have been detected in patients with GD. The actions of these antibodies initiate signaling in orbital fibroblasts from patients with the disease. Further, we have identified a functional and physical interaction between TSHR and IGF1R. Importantly, it appears that signaling initiated from either receptor can be attenuated by inhibiting the activity of IGF1R. These findings underpin the rationale for therapeutically targeting IGF1R in active TAO. A recently completed therapeutic trial of teprotumumab, a human IGF1R inhibiting antibody, in patients with moderate to severe, active TAO, indicates the potential effectiveness and safety of the drug. It is possible that other autoimmune diseases might also benefit from this treatment strategy.
Keywords: IGF receptor; immune system; signal transduction; thyroid.
© 2018 Society for Endocrinology.
Conflict of interest statement
Declaration of Interests
The author has been issued patents while a faculty member at the UCLA School of Medicine covering the diagnostic methods for monitoring anti-IGF-IR antibodies and the therapeutic targeting of IGF-IR in Graves’ disease and other autoimmune diseases. I have requested that my current employer, the University of Michigan Medical School, adjudicate any conflicts of interest.
Figures

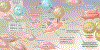

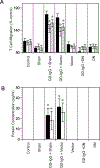



References
-
- Bahn RS, Dutton CM, Natt N, Joba W, Spitzweg C & Heufelder AE 1998. Thyrotropin receptor expression in Graves’ orbital adipose/connective tissues: potential autoantigen in Graves’ ophthalmopathy. J Clin Endocrinol Metab 83 998–1002. - PubMed
-
- Baldeschi L, Lupetti A, Vu P, Wakelkamp IM, Prummel MF & Wiersinga WM 2007. Reactivation of Graves’ orbitopathy after rehabilitative orbital decompression. Ophthalmology 114 1395–1402. - PubMed
-
- Barrett JP, Minogue AM, Falvey A & Lynch MA 2015. Involvement of IGF-1 and Akt in M1/M2 activation state in bone marrow-derived macrophages. Exp Cell Res 335 258–268. - PubMed
-
- Bartalena L, Baldeschi L, Boboridis K, Eckstein A, Kahaly GJ, Marcocci C, Perros P, Salvi M, Wiersinga WM & European Group on Graves O 2016. The 2016 European Thyroid Association/European Group on Graves’ Orbitopathy Guidelines for the Management of Graves’ Orbitopathy. Eur Thyroid J 5 9–26. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

