Generation and Functional In Vitro Analysis of Semliki Forest Virus Vectors Encoding TNF-α and IFN-γ
- PMID: 29276511
- PMCID: PMC5727424
- DOI: 10.3389/fimmu.2017.01667
Generation and Functional In Vitro Analysis of Semliki Forest Virus Vectors Encoding TNF-α and IFN-γ
Abstract
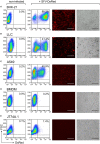
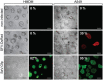
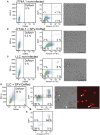
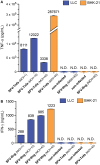
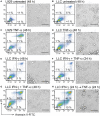
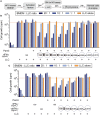
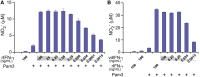
Cytokine gene delivery by viral vectors is a promising novel strategy for cancer immunotherapy. Semliki Forest virus (SFV) has many advantages as a delivery vector, including the ability to (i) induce p53-independent killing of tumor cells via apoptosis, (ii) elicit a type-I interferon (IFN) response, and (iii) express high levels of the transgene. SFV vectors encoding cytokines such as interleukin (IL)-12 have shown promising therapeutic responses in experimental tumor models. Here, we developed two new recombinant SFV vectors encoding either murine tumor necrosis factor-α (TNF-α) or murine interferon-γ (IFN-γ), two cytokines with documented immunostimulatory and antitumor activity. The SFV vector showed high infection rate and cytotoxicity in mouse and human lung carcinoma cells in vitro. By contrast, mouse and human macrophages were resistant to infection with SFV. The recombinant SFV vectors directly inhibited mouse lung carcinoma cell growth in vitro, while exploiting the cancer cells for production of SFV vector-encoded cytokines. The functionality of SFV vector-derived TNF-α was confirmed through successful induction of cell death in TNF-α-sensitive fibroblasts in a concentration-dependent manner. SFV vector-derived IFN-γ activated macrophages toward a tumoricidal phenotype leading to suppressed Lewis lung carcinoma cell growth in vitro in a concentration-dependent manner. The ability of SFV to provide functional cytokines and infect tumor cells but not macrophages suggests that SFV may be very useful for cancer immunotherapy employing tumor-infiltrating macrophages.
Keywords: Lewis lung carcinoma; Semliki Forest virus; cancer immunotherapy; cytokines; gene delivery; macrophage activation.
Figures



Similar articles
-
Macrophage Transcriptomic Alterations Driven by Alphavirus-Based Cancer Immunotherapy Vectors.J Immunol Res. 2025 Jun 13;2025:6573891. doi: 10.1155/jimr/6573891. eCollection 2025. J Immunol Res. 2025. PMID: 40547518 Free PMC article.
-
A Semliki forest virus vector engineered to express IFNα induces efficient elimination of established tumors.Gene Ther. 2012 Mar;19(3):271-8. doi: 10.1038/gt.2011.99. Epub 2011 Jul 7. Gene Ther. 2012. PMID: 21734727
-
Role of interferon-gamma in T-cell responses to Semliki Forest virus-infected murine brain cells.Immunology. 1988 Mar;63(3):355-62. Immunology. 1988. PMID: 2832313 Free PMC article.
-
Gamma interferon production and cytotoxicity of spleen cells from mice infected with Semliki Forest virus.J Gen Virol. 1984 May;65 ( Pt 5):955-61. doi: 10.1099/0022-1317-65-5-955. J Gen Virol. 1984. PMID: 6327889
-
Alphaviruses and their derived vectors as anti-tumor agents.Curr Cancer Drug Targets. 2004 Nov;4(7):597-607. doi: 10.2174/1568009043332754. Curr Cancer Drug Targets. 2004. PMID: 15578917 Review.
Cited by
-
Recombinant Virus Quantification Using Single-Cell Droplet Digital PCR: A Method for Infectious Titer Quantification.Viruses. 2023 Apr 26;15(5):1060. doi: 10.3390/v15051060. Viruses. 2023. PMID: 37243145 Free PMC article.
-
Targeting the Blood-Brain Tumor Barrier with Tumor Necrosis Factor-α.Pharmaceutics. 2022 Jul 6;14(7):1414. doi: 10.3390/pharmaceutics14071414. Pharmaceutics. 2022. PMID: 35890309 Free PMC article. Review.
-
Recombinant Viral Vectors for Therapeutic Programming of Tumour Microenvironment: Advantages and Limitations.Biomedicines. 2022 Aug 31;10(9):2142. doi: 10.3390/biomedicines10092142. Biomedicines. 2022. PMID: 36140243 Free PMC article. Review.
-
Both Type I and Type II Interferons Can Activate Antitumor M1 Macrophages When Combined With TLR Stimulation.Front Immunol. 2018 Nov 2;9:2520. doi: 10.3389/fimmu.2018.02520. eCollection 2018. Front Immunol. 2018. PMID: 30450098 Free PMC article.
-
Styrylpyridinium Derivatives for Fluorescent Cell Imaging.Pharmaceuticals (Basel). 2023 Sep 4;16(9):1245. doi: 10.3390/ph16091245. Pharmaceuticals (Basel). 2023. PMID: 37765053 Free PMC article.
References
-
- Lundstrom K. Viral vector-based cancer immunotherapy. Austin Immunol (2016) 1(2):1008.
-
- Asselin-Paturel C, Lassau N, Guinebretière JM, Zhang J, Gay F, Bex F, et al. Transfer of the murine interleukin-12 gene in vivo by a Semliki Forest virus vector induces B16 tumor regression through inhibition of tumor blood vessel formation monitored by Doppler ultrasonography. Gene Ther (1999) 6(4):606–15.10.1038/sj.gt.3300841 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

