Cellulose synthase complex organization and cellulose microfibril structure
- PMID: 29277745
- PMCID: PMC5746560
- DOI: 10.1098/rsta.2017.0048
Cellulose synthase complex organization and cellulose microfibril structure
Abstract
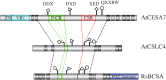
Cellulose consists of linear chains of β-1,4-linked glucose units, which are synthesized by the cellulose synthase complex (CSC). In plants, these chains associate in an ordered manner to form the cellulose microfibrils. Both the CSC and the local environment in which the individual chains coalesce to form the cellulose microfibril determine the structure and the unique physical properties of the microfibril. There are several recent reviews that cover many aspects of cellulose biosynthesis, which include trafficking of the complex to the plasma membrane and the relationship between the movement of the CSC and the underlying cortical microtubules (Bringmann et al. 2012 Trends Plant Sci.17, 666-674 (doi:10.1016/j.tplants.2012.06.003); Kumar & Turner 2015 Phytochemistry112, 91-99 (doi:10.1016/j.phytochem.2014.07.009); Schneider et al. 2016 Curr. Opin. Plant Biol.34, 9-16 (doi:10.1016/j.pbi.2016.07.007)). In this review, we will focus on recent advances in cellulose biosynthesis in plants, with an emphasis on our current understanding of the structure of individual catalytic subunits together with the local membrane environment where cellulose synthesis occurs. We will attempt to relate this information to our current knowledge of the structure of the cellulose microfibril and propose a model in which variations in the structure of the CSC have important implications for the structure of the cellulose microfibril produced.This article is part of a discussion meeting issue 'New horizons for cellulose nanotechnology'.
Keywords: cellulose; electron microscopy; microfibril; protein complex; synthesis.
© 2017 The Author(s).
Conflict of interest statement
We declare we have no competing interests.
Figures


Similar articles
-
Structure of native cellulose microfibrils, the starting point for nanocellulose manufacture.Philos Trans A Math Phys Eng Sci. 2018 Feb 13;376(2112):20170045. doi: 10.1098/rsta.2017.0045. Philos Trans A Math Phys Eng Sci. 2018. PMID: 29277742 Review.
-
Plant cellulose synthesis: CESA proteins crossing kingdoms.Phytochemistry. 2015 Apr;112:91-9. doi: 10.1016/j.phytochem.2014.07.009. Epub 2014 Aug 4. Phytochemistry. 2015. PMID: 25104231 Review.
-
Cellulose microfibrils in plants: biosynthesis, deposition, and integration into the cell wall.Int Rev Cytol. 2000;199:161-99. doi: 10.1016/s0074-7696(00)99004-1. Int Rev Cytol. 2000. PMID: 10874579 Review.
-
Cellulose microfibril assembly and orientation: recent developments.J Cell Sci Suppl. 1985;2:13-32. doi: 10.1242/jcs.1985.supplement_2.2. J Cell Sci Suppl. 1985. PMID: 3867669
-
Cellulose microfibril alignment recovers from DCB-induced disruption despite microtubule disorganization.Plant J. 2003 Nov;36(4):565-75. doi: 10.1046/j.1365-313x.2003.01906.x. Plant J. 2003. PMID: 14617086
Cited by
-
Polysaccharide Biosynthesis: Glycosyltransferases and Their Complexes.Front Plant Sci. 2021 Feb 19;12:625307. doi: 10.3389/fpls.2021.625307. eCollection 2021. Front Plant Sci. 2021. PMID: 33679837 Free PMC article.
-
The Cytoskeleton and Its Role in Determining Cellulose Microfibril Angle in Secondary Cell Walls of Woody Tree Species.Plants (Basel). 2020 Jan 10;9(1):90. doi: 10.3390/plants9010090. Plants (Basel). 2020. PMID: 31936868 Free PMC article. Review.
-
Raman imaging of Micrasterias: new insights into shape formation.Protoplasma. 2021 Nov;258(6):1323-1334. doi: 10.1007/s00709-021-01685-3. Epub 2021 Jul 22. Protoplasma. 2021. PMID: 34292402 Free PMC article.
-
Recent advances in enzymatic synthesis of β-glucan and cellulose.Carbohydr Res. 2021 Oct;508:108411. doi: 10.1016/j.carres.2021.108411. Epub 2021 Jul 24. Carbohydr Res. 2021. PMID: 34392134 Free PMC article. Review.
-
CRISPR/Cas9-mediated P-CR domain-specific engineering of CESA4 heterodimerization capacity alters cell wall architecture and improves saccharification efficiency in poplar.Plant Biotechnol J. 2022 Jun;20(6):1197-1212. doi: 10.1111/pbi.13803. Epub 2022 Mar 15. Plant Biotechnol J. 2022. PMID: 35266285 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

