Comparison of reverse-transcription real-time PCR and immunohistochemistry for the detection of canine distemper virus infection in raccoons in Ontario, Canada
- PMID: 29284384
- PMCID: PMC6505874
- DOI: 10.1177/1040638717751825
Comparison of reverse-transcription real-time PCR and immunohistochemistry for the detection of canine distemper virus infection in raccoons in Ontario, Canada
Abstract
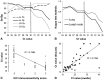
Canine distemper virus (CDV) is a widespread morbillivirus that causes subclinical to fatal infections in domestic and wild carnivores. Raccoons ( Procyon lotor) are CDV reservoirs and suffer from associated disease. Aspects of pathogenesis may lead to difficulty in the interpretation of commonly used testing modalities, such as reverse-transcription real-time (RT-rt)PCR and immunohistochemistry (IHC). The reliance upon such tests is greater for wildlife, which are often submitted as carcasses with no clinical history. We compared CDV RT-rtPCR results to immunohistochemistry (the gold standard) in tissues from 74 raccoons. These tests had high kappa agreement (lymph node: 0.9335; lung: 0.8671) and a negative correlation between IHC score and threshold cycle (Ct) value for lymph node and lung (Spearman rank correlation coefficient [ rs] = -0.8555 and -0.8179, respectively; p < 0.00001). An RT-rtPCR Ct value of 30 in lung and lymph node with sensitivity and specificity of 92.3 and 92.6% and 86.8 and 96.4%, respectively, was suitable for determining CDV involvement. Conjunctival swabs provide an alternative for distemper diagnosis, as there was a strong correlation between Ct values of conjunctival swabs and tissues ( rs = -0.8498, p < 0.00001, n = 46). This information will aid in more efficient and accurate diagnoses in individuals, small-scale outbreaks, and epidemiologic investigations in wildlife.
Keywords: Detection test; distemper; morbillivirus; raccoon; wild animal.
Conflict of interest statement
Figures
Similar articles
-
Characterization of a novel Canine distemper virus causing disease in wildlife.J Vet Diagn Invest. 2016 Sep;28(5):506-13. doi: 10.1177/1040638716656025. Epub 2016 Jul 7. J Vet Diagn Invest. 2016. PMID: 27400957
-
Canine distemper outbreak in raccoons suggests pathogen interspecies transmission amongst alien and native carnivores in urban areas from Germany.Vet Microbiol. 2014 Nov 7;174(1-2):50-9. doi: 10.1016/j.vetmic.2014.08.034. Epub 2014 Sep 10. Vet Microbiol. 2014. PMID: 25258173
-
Frequency of Virus Coinfection in Raccoons ( Procyon lotor) and Striped Skunks ( Mephitis mephitis) During a Concurrent Rabies and Canine Distemper Outbreak.J Wildl Dis. 2018 Jul;54(3):622-625. doi: 10.7589/2017-04-072. Epub 2018 Mar 8. J Wildl Dis. 2018. PMID: 29517402
-
Advances in canine distemper virus pathogenesis research: a wildlife perspective.J Gen Virol. 2017 Mar;98(3):311-321. doi: 10.1099/jgv.0.000666. Epub 2017 Apr 1. J Gen Virol. 2017. PMID: 27902345 Review.
-
[Canine distemper virus--an agent looking for new hosts].Dtsch Tierarztl Wochenschr. 2003 Apr;110(4):137-42. Dtsch Tierarztl Wochenschr. 2003. PMID: 12756952 Review. German.
Cited by
-
Development of a double monoclonal antibody-based sandwich enzyme-linked immunosorbent assay for detecting canine distemper virus.Appl Microbiol Biotechnol. 2020 Dec;104(24):10725-10735. doi: 10.1007/s00253-020-10997-y. Epub 2020 Nov 7. Appl Microbiol Biotechnol. 2020. PMID: 33159543 Free PMC article.
-
Bacterial and Viral Pathogens with One Health Relevance in Invasive Raccoons (Procyon lotor, Linné 1758) in Southwest Germany.Pathogens. 2023 Mar 1;12(3):389. doi: 10.3390/pathogens12030389. Pathogens. 2023. PMID: 36986312 Free PMC article.
References
-
- Appel MJG, et al. Canine distemper virus induced infection and encephalitis in javelinas (collared peccaries). Arch Virol 1991;119:147–152. - PubMed
-
- Appel MJG, Summers BA. Pathogenicity of morbilliviruses for terrestrial carnivores. Vet Microbiol 1995;44:187–191. - PubMed
-
- Beineke A, et al. Pathogenesis and immunopathology of systemic and nervous canine distemper. Vet Immunol Immunopathol 2009;127:1–18. - PubMed
-
- Caraguel CGB, et al. Selection of a cutoff value for real-time polymerase chain reaction results to fit a diagnostic purpose: analytical and epidemiologic approaches. J Vet Diagn Invest 2011;23:2–15. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

