Active maintenance of endothelial cells prevents kidney fibrosis
- PMID: 29285425
- PMCID: PMC5743042
- DOI: 10.23876/j.krcp.2017.36.4.329
Active maintenance of endothelial cells prevents kidney fibrosis
Abstract
Background: Soluble epoxide hydrolase (sEH) expressed by endothelial cells catalyzes the metabolism of epoxyeicosatrienoic acids (EETs), which are vasoactive agents.
Methods: We used a unilateral ureteral obstruction mouse model of kidney fibrosis to determine whether inhibition of sEH activity reduces fibrosis, the final common pathway for chronic kidney disease.
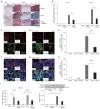
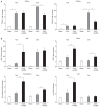
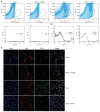
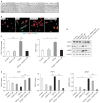
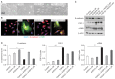
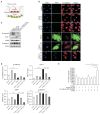
Results: sEH activity was inhibited by continuous release of the inhibitor 12-(3-adamantan-1-ylureido)-dodecanoic acid (AUDA) for 1 or 2 weeks. Treatment with AUDA significantly ameliorated tubulointerstitial fibrosis by reducing fibroblast mobilization and enhancing endothelial cell activity. In an in vitro model of endothelial-to-mesenchymal transition (EndMT) using human vascular endothelial cells (HUVECs), AUDA prevented the morphologic changes associated with EndMT and reduced expression of fibroblast-specific protein 1. Furthermore, HUVECs activated by AUDA prevented the epithelial-to-mesenchymal transition (EMT) of tubular epithelial cells in a co-culture system.
Conclusion: Our findings suggest that regulation of sEH is a potential target for therapies aimed at delaying the progression of kidney fibrosis by inhibiting EndMT and EMT.
Keywords: Endothelial dysfunction; Endothelial-to-mesenchymal transition; Epithelial-mesenchymal transition; Kidney fibrosis; Soluble epoxide hydrolase.
Conflict of interest statement
Conflicts of interest All authors have no conflicts of interest to declare.
Figures
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

