Implementation of "clinical sequencing" in cancer genome medicine in Japan
- PMID: 29285848
- PMCID: PMC5834803
- DOI: 10.1111/cas.13486
Implementation of "clinical sequencing" in cancer genome medicine in Japan
Abstract
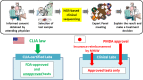
In oncology, actionable mutations (alterations) in cancer-associated genes are critical in terms of the selection of therapeutic approaches. Next-generation sequencing of tumor sample DNA (ie, clinical sequencing) can guide clinical management by providing diagnostic or prognostic data, and facilitating the identification of potential treatment regimens, such as molecular-targeted and immune checkpoint blockade therapies. In the USA, a variety of tumor-profiling multiplex gene panels have been developed and implemented for this purpose. In Japan, several academic institutions have now carried out detailed investigations of the feasibility and value of clinical sequencing, and cancer societies have issued consensus clinical practice guidance for next-generation sequencing-based gene panel tests. These efforts will facilitate the implementation of cancer genome medicine in Japan.
Keywords: actionable mutation; cancer genome medicine; clinical sequencing; implementation; insurance reimbursement.
© 2018 The Authors. Cancer Science published by John Wiley & Sons Australia, Ltd on behalf of Japanese Cancer Association.
Figures

References
-
- Carr TH, McEwen R, Dougherty B, et al. Defining actionable mutations for oncology therapeutic development. Nat Rev Cancer. 2016;16:319‐329. - PubMed
-
- Yoh K, Seto T, Satouchi M, et al. Vandetanib in patients with previously treated RET‐rearranged advanced non‐small‐cell lung cancer (LURET): an open‐label, multicentre phase 2 trial. Lancet Respir Med. 2017;5:42‐50. - PubMed
-
- Planchard D, Smit EF, Groen HJM, et al. Dabrafenib plus trametinib in patients with previously untreated BRAF(V600E)‐mutant metastatic non‐small‐cell lung cancer: an open‐label, phase 2 trial. Lancet Oncol. 2017;18:1307‐1316. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

