Reversible Conformational Conversion of α-Synuclein into Toxic Assemblies by Glucosylceramide
- PMID: 29290548
- PMCID: PMC6013314
- DOI: 10.1016/j.neuron.2017.12.012
Reversible Conformational Conversion of α-Synuclein into Toxic Assemblies by Glucosylceramide
Abstract
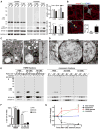
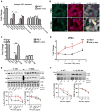
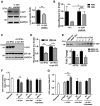
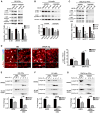
α-Synuclein (α-syn) aggregation is a key event in Parkinson's disease (PD). Mutations in glycosphingolipid (GSL)-degrading glucocerebrosidase are risk factors for PD, indicating that disrupted GSL clearance plays a key role in α-syn aggregation. However, the mechanisms of GSL-induced aggregation are not completely understood. We document the presence of physiological α-syn conformers in human midbrain dopamine neurons and tested their contribution to the aggregation process. Pathological α-syn assembly mainly occurred through the conversion of high molecular weight (HMW) physiological α-syn conformers into compact, assembly-state intermediates by glucosylceramide (GluCer), without apparent disassembly into free monomers. This process was reversible in vitro through GluCer depletion. Reducing GSLs in PD patient neurons with and without GBA1 mutations diminished pathology and restored physiological α-syn conformers that associated with synapses. Our work indicates that GSLs control the toxic conversion of physiological α-syn conformers in a reversible manner that is amenable to therapeutic intervention by GSL reducing agents.
Keywords: GBA1; Gaucher disease; Lewy body; Parkinson’s disease; glucocerebrosidase; iPS-derived dopaminergic neurons; lysosomal storage disease; protein aggregation; α-synuclein.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures



References
-
- Bessen RA, Kocisko DA, Raymond GJ, Nandan S, Lansbury PT, Caughey B. Non-genetic propagation of strain-specific properties of scrapie prion protein. Nature. 1995;375:698–700. - PubMed
-
- Bielawski J, Pierce JS, Snider J, Rembiesa B, Szulc ZM, Bielawska A. Sphingolipid analysis by high performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) Adv Exp Med Biol. 2010;688:46–59. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

