A negative allosteric modulator of PDE4D enhances learning after traumatic brain injury
- PMID: 29294383
- PMCID: PMC5844849
- DOI: 10.1016/j.nlm.2017.12.008
A negative allosteric modulator of PDE4D enhances learning after traumatic brain injury
Abstract
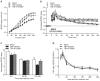
Traumatic brain injury (TBI) significantly decreases cyclic AMP (cAMP) signaling which produces long-term synaptic plasticity deficits and chronic learning and memory impairments. Phosphodiesterase 4 (PDE4) is a major family of cAMP hydrolyzing enzymes in the brain and of the four PDE4 subtypes, PDE4D in particular has been found to be involved in memory formation. Although most PDE4 inhibitors target all PDE4 subtypes, PDE4D can be targeted with a selective, negative allosteric modulator, D159687. In this study, we hypothesized that treating animals with D159687 could reverse the cognitive deficits caused by TBI. To test this hypothesis, adult male Sprague Dawley rats received sham surgery or moderate parasagittal fluid-percussion brain injury. After 3 months of recovery, animals were treated with D159687 (0.3 mg/kg, intraperitoneally) at 30 min prior to cue and contextual fear conditioning, acquisition in the water maze or during a spatial working memory task. Treatment with D159687 had no significant effect on these behavioral tasks in non-injured, sham animals, but did reverse the learning and memory deficits in chronic TBI animals. Assessment of hippocampal slices at 3 months post-TBI revealed that D159687 reversed both the depression in basal synaptic transmission in area CA1 as well as the late-phase of long-term potentiation. These results demonstrate that a negative allosteric modulator of PDE4D may be a potential therapeutic to improve chronic cognitive dysfunction following TBI.
Keywords: Cognition; Fluid Percussion Injury; Hippocampus; Memory; Phosphodiesterase; Traumatic brain injury.
Copyright © 2018 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

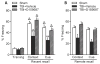
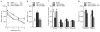
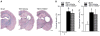


References
-
- Barad M, Bourtchouladze R, Winder DG, Golan H, Kandel E. Rolipram, a type IV-specific phosphodiesterase inhibitor, facilitates the establishment of long-lasting long-term potentiation and improves memory. Proceedings of the National Academy of Sciences of the United States of America. 1998;95:15020–15025. - PMC - PubMed
-
- Bourtchuladze R, Frenguelli B, Blendy J, Cioffi D, Schutz G, Silva AJ. Deficient long-term memory in mice with a targeted mutation of the cAMP-responsive element-binding protein. Cell. 1994;79:59–68. - PubMed
-
- Brullo C, Ricciarelli R, Prickaerts J, Arancio O, Massa M, Rotolo C, Romussi A, Rebosio C, Marengo B, Pronzato MA, van Hagen BT, van Goethem NP, D’Ursi P, Orro A, Milanesi L, Guariento S, Cichero E, Fossa P, Fedele E, Bruno O. New insights into selective PDE4D inhibitors: 3-(Cyclopentyloxy)-4-methoxybenzaldehyde O-(2-(2,6-dimethylmorpholino)-2-oxoethyl) oxime (GEBR-7b) structural development and promising activities to restore memory impairment. Eur J Med Chem. 2016;124:82–102. - PubMed
-
- Bruno O, Fedele E, Prickaerts J, Parker LA, Canepa E, Brullo C, Cavallero A, Gardella E, Balbi A, Domenicotti C, Bollen E, Gijselaers HJ, Vanmierlo T, Erb K, Limebeer CL, Argellati F, Marinari UM, Pronzato MA, Ricciarelli R. GEBR-7b, a novel PDE4D selective inhibitor that improves memory in rodents at non-emetic doses. British Journal of Pharmacology. 2011;164:2054–2063. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

