Fibrin and D-dimer bind to monomeric GPVI
- PMID: 29296791
- PMCID: PMC5728468
- DOI: 10.1182/bloodadvances.2017007732
Fibrin and D-dimer bind to monomeric GPVI
Abstract
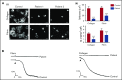
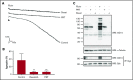
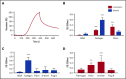
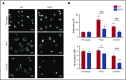
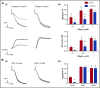
Fibrin has recently been shown to activate platelets through the immunoglobulin receptor glycoprotein VI (GPVI). In the present study, we show that spreading of human platelets on fibrin is abolished in patients deficient in GPVI, confirming that fibrin activates human platelets through the immunoglobulin receptor. Using a series of proteolytic fragments, we show that D-dimer, but not the E fragment of fibrin, binds to GPVI and that immobilized D-dimer induces platelet spreading through activation of Src and Syk tyrosine kinases. In contrast, when platelets are activated in suspension, soluble D-dimer inhibits platelet aggregation induced by fibrin and collagen, but not by a collagen-related peptide composed of a repeat GPO sequence or by thrombin. Using surface plasmon resonance, we demonstrate that fibrin binds selectively to monomeric GPVI with a KD of 302 nM, in contrast to collagen, which binds primarily to dimeric GPVI. These results establish GPVI as the major signaling receptor for fibrin in human platelets and provide evidence that fibrin binds to a distinct configuration of GPVI. This indicates that it may be possible to develop agents that selectively block the interaction of fibrin but not collagen with the immunoglobulin receptor. Such agents are required to establish whether selective targeting of either interaction has the potential to lead to development of an antithrombotic agent with a reduced effect on bleeding relative to current antiplatelet drugs.
Conflict of interest statement
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Figures


References
-
- Mammadova-Bach E, Ollivier V, Loyau S, et al. Platelet glycoprotein VI binds to polymerized fibrin and promotes thrombin generation. Blood. 2015;126(5):683-691. - PubMed
-
- Bender M, Hagedorn I, Nieswandt B. Genetic and antibody-induced glycoprotein VI deficiency equally protects mice from mechanically and FeCl(3) -induced thrombosis. J Thromb Haemost. 2011;9(7):1423-1426. - PubMed
-
- Matus V, Valenzuela G, Sáez CG, et al. An adenine insertion in exon 6 of human GP6 generates a truncated protein associated with a bleeding disorder in four Chilean families. J Thromb Haemost. 2013;11(9):1751-1759. - PubMed
-
- Jiang P, Jandrot-Perrus M. New advances in treating thrombotic diseases: GPVI as a platelet drug target. Drug Discov Today. 2014;19(9):1471-1475. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

