Cell-surface C-type lectin-like receptor CLEC-1 dampens dendritic cell activation and downstream Th17 responses
- PMID: 29296975
- PMCID: PMC5728597
- DOI: 10.1182/bloodadvances.2016002360
Cell-surface C-type lectin-like receptor CLEC-1 dampens dendritic cell activation and downstream Th17 responses
Abstract
Dendritic cells (DCs) represent essential antigen-presenting cells that are critical for linking innate and adaptive immunity, and influencing T-cell responses. Among pattern recognition receptors, DCs express C-type lectin receptors triggered by both exogenous and endogenous ligands, therefore dictating pathogen response, and also shaping T-cell immunity. We previously described in rat, the expression of the orphan C-type lectin-like receptor-1 (CLEC-1) by DCs and demonstrated in vitro its inhibitory role in downstream T helper 17 (Th17) activation. In this study, we examined the expression and functionality of CLEC-1 in human DCs, and show a cell-surface expression on the CD16- subpopulation of blood DCs and on monocyte-derived DCs (moDCs). CLEC-1 expression on moDCs is downregulated by inflammatory stimuli and enhanced by transforming growth factor β. Moreover, we demonstrate that CLEC-1 is a functional receptor on human moDCs and that although not modulating the spleen tyrosine kinase-dependent canonical nuclear factor-κB pathway, represses subsequent Th17 responses. Interestingly, a decreased expression of CLEC1A in human lung transplants is predictive of the development of chronic rejection and is associated with a higher level of interleukin 17A (IL17A). Importantly, using CLEC-1-deficient rats, we showed that disruption of CLEC-1 signaling led to an enhanced Il12p40 subunit expression in DCs, and to an exacerbation of downstream in vitro and in vivo CD4+ Th1 and Th17 responses. Collectively, our results establish a role for CLEC-1 as an inhibitory receptor in DCs able to dampen activation and downstream effector Th responses. As a cell-surface receptor, CLEC-1 may represent a useful therapeutic target for modulating T-cell immune responses in a clinical setting.
Conflict of interest statement
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Figures

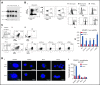


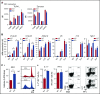

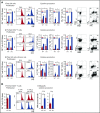
References
-
- Thebault P, Lhermite N, Tilly G, et al. . The C-type lectin-like receptor CLEC-1, expressed by myeloid cells and endothelial cells, is up-regulated by immunoregulatory mediators and moderates T cell activation. J Immunol. 2009;183(5):3099-3108. - PubMed
-
- Colonna M, Samaridis J, Angman L. Molecular characterization of two novel C-type lectin-like receptors, one of which is selectively expressed in human dendritic cells. Eur J Immunol. 2000;30(2):697-704. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

