Effectiveness and safety of ferric carboxymaltose compared to iron sucrose in women with iron deficiency anemia: phase IV clinical trials
- PMID: 29304848
- PMCID: PMC5755312
- DOI: 10.1186/s12905-017-0506-8
Effectiveness and safety of ferric carboxymaltose compared to iron sucrose in women with iron deficiency anemia: phase IV clinical trials
Abstract
Background: Iron deficiency anemia (IDA) is a significant problem worldwide particularly in women. The aim of the study was to evaluate the effectiveness and safety of intravenous ferric carboxymaltose (FCM) in comparison to iron sucrose (IS) in women with IDA.
Method: Two hundred patients at Department of Obstetrics and Gynaecology, Sher-i-Kashmir Institute of Medical Sciences Medical College and Hospital, Jammu & Kashmir, India identified with IDA were enrolled for the study. Intravenous FCM and IS were both given as per the protocol. Change in the laboratory parameters such as hemoglobin (Hb), mean corpuscular value, and serum ferritin levels at two weeks and four weeks interval after the treatment was recorded.
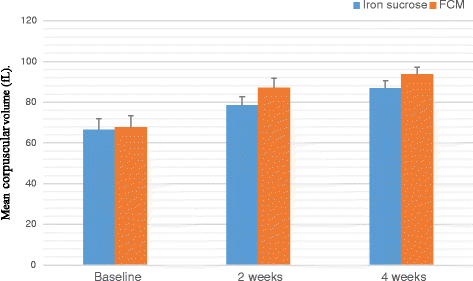
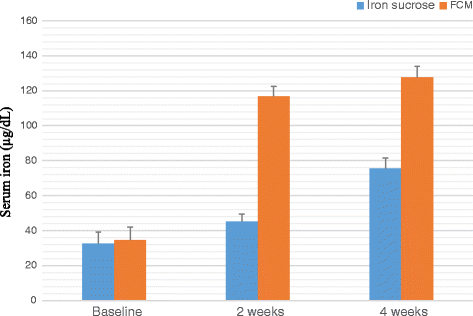
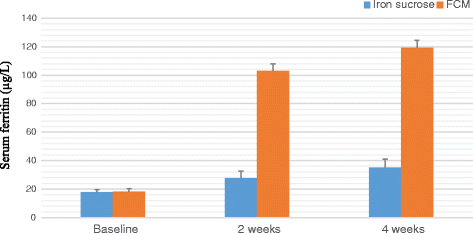
Result: A significant increase in the mean Hb was observed from 7.76 ± 0.709 to 13.25 ± 0.606 in patients treated with FCM and 7.64 ± 0.710 to 11.59 ± 0.733 g/dL (P < 0.001) in patients treated with IS after four weeks of therapy. The rise in mean corpuscular volume was from 66.82 ± 5.24 to 86.76 ± 3.765 and 68.05 ± 5.56 to 93.80 ± 3.80 and rise in serum ferritin levels were from 8.32 ± 1.787 to 38.94 ± 6.095 μg/L and 8.16 ± 1.540 to 27 ± 8.175 μg/L in patients treated with FCM and IS respectively after four weeks of therapy. No serious adverse effects were reported.
Conclusion: Parenteral therapy is effective in IDA, but FCM elevates hemoglobin level and restored iron stores faster than IS with minimum adverse drug reactions.
Trial registration number: ISRCTN14484575 Dated: 15-12-2017 retrospectively registered. https://doi.org/10.1186/ISRCTN14484575.
Keywords: Ferric carboxymaltose; Hemoglobin; Iron deficiency anemia; Iron sucrose; Serum ferritin.
Conflict of interest statement
Ethics approval and consent to participate
Institutional Ethics Committee Sher-i-Kashmir Institute of Medical Sciences Medical College and Hospital (IEC - SKIMS MCH), in accordance with Indian Council of Medical Research (ICMR) guidelines. Institutional Ethical Registration Number: SKIMS MC/CM/IEC/15/42–50. Dated 16–07-2015.
Written as well as verbal consent was taken by each participant.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures




Similar articles
-
Comparison of ferric Carboxymaltose and iron sucrose complex for treatment of iron deficiency anemia in pregnancy- randomised controlled trial.BMC Pregnancy Childbirth. 2019 Feb 4;19(1):54. doi: 10.1186/s12884-019-2200-3. BMC Pregnancy Childbirth. 2019. PMID: 30717690 Free PMC article. Clinical Trial.
-
Comparative efficacy and safety of intravenous ferric carboxymaltose and iron sucrose for iron deficiency anemia in obstetric and gynecologic patients: A systematic review and meta-analysis.Medicine (Baltimore). 2021 May 21;100(20):e24571. doi: 10.1097/MD.0000000000024571. Medicine (Baltimore). 2021. PMID: 34011020 Free PMC article.
-
Efficacy and cost effectiveness of intravenous ferric carboxymaltose versus iron sucrose in adult patients with iron deficiency anaemia.PLoS One. 2021 Aug 10;16(8):e0255104. doi: 10.1371/journal.pone.0255104. eCollection 2021. PLoS One. 2021. PMID: 34375369 Free PMC article.
-
TIDILAP: Treatment of iron deficiency in lipoprotein apheresis patients --A prospective observational multi-center cohort study comparing efficacy, safety and tolerability of ferric gluconate with ferric carboxymaltose.Atheroscler Suppl. 2015 May;18:199-208. doi: 10.1016/j.atherosclerosissup.2015.02.030. Atheroscler Suppl. 2015. PMID: 25936327
-
Efficacy, Safety, and Tolerability of Ferric Carboxymaltose and Iron Sucrose in Iron-Deficiency Anemia: A Systematic Review and Meta-Analysis of Randomized Controlled Trials.Turk J Haematol. 2025 May 22;42(2):119-135. doi: 10.4274/tjh.galenos.2025.2025.0034. Epub 2025 Mar 17. Turk J Haematol. 2025. PMID: 40091548 Free PMC article.
Cited by
-
Evaluation of a treatment protocol for anaemia in pregnancy nested in routine antenatal care in a limited-resource setting.Glob Health Action. 2019;12(1):1621589. doi: 10.1080/16549716.2019.1621589. Glob Health Action. 2019. PMID: 31203791 Free PMC article.
-
Ferric Carboxymaltose in the Management of Iron Deficiency Anemia in Pregnancy: A Subgroup Analysis of a Multicenter Real-World Study Involving 1191 Pregnant Women.Obstet Gynecol Int. 2022 Nov 28;2022:5759740. doi: 10.1155/2022/5759740. eCollection 2022. Obstet Gynecol Int. 2022. PMID: 36479303 Free PMC article.
-
Efficacy and Safety of Ferric Carboxymaltose in the Management of Iron Deficiency Anemia: A Multi-Center Real-World Study from India.J Blood Med. 2022 Jun 8;13:303-313. doi: 10.2147/JBM.S361210. eCollection 2022. J Blood Med. 2022. PMID: 35706850 Free PMC article.
-
REGAIN STUDY: Retrospective Study to Assess the Effectiveness, Tolerability, and Safety of Ferric Carboxymaltose in the Management of Iron Deficiency Anemia in Pregnant Women.Anemia. 2019 Nov 12;2019:4640635. doi: 10.1155/2019/4640635. eCollection 2019. Anemia. 2019. PMID: 31781389 Free PMC article.
-
Ferric carboxymaltose: A game changer in the management of iron deficiency anaemia in pregnancy.J Family Med Prim Care. 2024 Jun;13(6):2379-2384. doi: 10.4103/jfmpc.jfmpc_1258_23. Epub 2024 Jun 14. J Family Med Prim Care. 2024. PMID: 39027861 Free PMC article.
References
-
- Glazer Y, Bilenko N. Effect of iron deficiency and iron deficiency anemia in the first two years of life on cognitive and mental development during childhood. Harefuah. 2010;149(5):309–314,335. - PubMed
-
- WHO. Nutritional anemias. Report of a WHO scientific group. Geneva, World Health Organization, 1968. (WHO Technical Report Series, No. 405). http://apps.who.int/iris/bitstream/10665/40707/1/WHO_TRS_405.pdf. Accessed 5 Nov 2017. - PubMed
-
- WHO/UNICEF/UNU. Iron deficiency anaemia assessment, prevention, and control: a guide for programme managers. Geneva, World Health Organization, 2001. Available at: http://www.who.int/nutrition/publications/micronutrients/anaemia_iron_de.... Accessed 5 Nov 2017.
-
- Global Burden of Disease Study 2013, Collaborators Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the global burden of disease study 2013. Lancet. 2015;386(9995):743–800. doi: 10.1016/S0140-6736(15)60692-4. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources

