An Oxygen-Tolerant PET-RAFT Polymerization for Screening Structure-Activity Relationships
- PMID: 29316089
- PMCID: PMC9641662
- DOI: 10.1002/anie.201711044
An Oxygen-Tolerant PET-RAFT Polymerization for Screening Structure-Activity Relationships
Abstract
The complexity of polymer-protein interactions makes rational design of the best polymer architecture for any given biointerface extremely challenging, and the high throughput synthesis and screening of polymers has emerged as an attractive alternative. A porphyrin-catalysed photoinduced electron/energy transfer-reversible addition-fragmentation chain-transfer (PET-RAFT) polymerisation was adapted to enable high throughput synthesis of complex polymer architectures in dimethyl sulfoxide (DMSO) on low-volume well plates in the presence of air. The polymerisation system shows remarkable oxygen tolerance, and excellent control of functional 3- and 4-arm star polymers. We then apply this method to investigate the effect of polymer structure on protein binding, in this case to the lectin concanavalin A (ConA). Such an approach could be applied to screen the structure-activity relationships for any number of polymer-protein interactions.
Keywords: PET-RAFT; click chemistry; glycopolymers; oxygen tolerance; photomediated radical polymerization.
© 2018 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
Conflict of interest
The authors declare no conflict of interest.
Figures
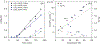

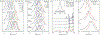
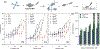
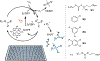
References
-
- Simon CG, Lin-Gibson S, Adv. Mater. 2011, 23, 369–387. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

