The Secretory Response of Rat Peritoneal Mast Cells on Exposure to Mineral Fibers
- PMID: 29320402
- PMCID: PMC5800203
- DOI: 10.3390/ijerph15010104
The Secretory Response of Rat Peritoneal Mast Cells on Exposure to Mineral Fibers
Abstract
Background: Exposure to mineral fibers is of substantial relevance to human health. A key event in exposure is the interaction with inflammatory cells and the subsequent generation of pro-inflammatory factors. Mast cells (MCs) have been shown to interact with titanium oxide (TiO₂) and asbestos fibers. In this study, we compared the response of rat peritoneal MCs challenged with the asbestos crocidolite and nanowires of TiO₂ to that induced by wollastonite employed as a control fiber.
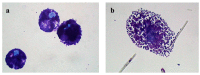
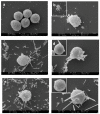
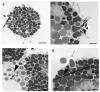
Methods: Rat peritoneal MCs (RPMCs), isolated from peritoneal lavage, were incubated in the presence of mineral fibers. The quantities of secreted enzymes were evaluated together with the activity of fiber-associated enzymes. The ultrastructural morphology of fiber-interacting RPMCs was analyzed with electron microscopy.
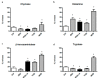
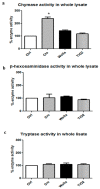
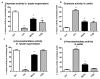
Results: Asbestos and TiO₂ stimulate MC secretion. Secreted enzymes bind to fibers and exhibit higher activity. TiO₂ and wollastonite bind and improve enzyme activity, but to a lesser degree than crocidolite.
Conclusions: (1) Mineral fibers are able to stimulate the mast cell secretory process by both active (during membrane interaction) and/or passive (during membrane penetration) interaction; (2) fibers can be found to be associated with secreted enzymes-this process appears to create long-lasting pro-inflammatory environments and may represent the active contribution of MCs in maintaining the inflammatory process; (3) MCs and their enzymes should be considered as a therapeutic target in the pathogenesis of asbestos-induced lung inflammation; and (4) MCs can contribute to the inflammatory effect associated with selected engineered nanomaterials, such as TiO₂ nanoparticles.
Keywords: asbestos; crocidolite; endocytosis; mast cells; secretory process; titanium oxide nanowires.
Conflict of interest statement
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
Figures



Similar articles
-
Mesothelial cell proliferation and biopersistence of wollastonite and crocidolite asbestos fibers.Fundam Appl Toxicol. 1997 Aug;38(2):173-83. doi: 10.1006/faat.1997.2344. Fundam Appl Toxicol. 1997. PMID: 9299191
-
Characterization of pulmonary responses in mice to asbestos/asbestiform fibers using gene expression profiles.J Toxicol Environ Health A. 2018;81(4):60-79. doi: 10.1080/15287394.2017.1408201. Epub 2017 Dec 26. J Toxicol Environ Health A. 2018. PMID: 29279043
-
Wollastonite fibers in vitro generate reactive oxygen species able to lyse erythrocytes and activate the complement alternate pathway.Toxicol Sci. 1998 Jul;44(1):32-8. doi: 10.1006/toxs.1998.2440. Toxicol Sci. 1998. PMID: 9720138
-
Mesothelioma and analysis of tissue fiber content.Recent Results Cancer Res. 2011;189:79-95. doi: 10.1007/978-3-642-10862-4_6. Recent Results Cancer Res. 2011. PMID: 21479897 Review.
-
[A case of pulmonary fibrosis with many asbestos bodies in bronchoalveolar lavage fluid after exposure to asbestos and man-made mineral fibers].Nihon Kokyuki Gakkai Zasshi. 2000 Oct;38(10):801-6. Nihon Kokyuki Gakkai Zasshi. 2000. PMID: 11186929 Review. Japanese.
Cited by
-
Mast Cells in Peritoneal Fluid From Women With Endometriosis and Their Possible Role in Modulating Sperm Function.Front Physiol. 2020 Jan 9;10:1543. doi: 10.3389/fphys.2019.01543. eCollection 2019. Front Physiol. 2020. PMID: 31998139 Free PMC article.
-
Autoantibodies and cancer among asbestos-exposed cohorts in Western Australia.J Toxicol Environ Health A. 2021 Jun 3;84(11):475-483. doi: 10.1080/15287394.2021.1889424. Epub 2021 Mar 7. J Toxicol Environ Health A. 2021. PMID: 33678145 Free PMC article.
-
On the mechanism of the electrophysiological changes and membrane lesions induced by asbestos fiber exposure in Xenopus laevis oocytes.Sci Rep. 2019 Feb 14;9(1):2014. doi: 10.1038/s41598-019-38591-x. Sci Rep. 2019. PMID: 30765791 Free PMC article.
-
Ferritin adsorption onto chrysotile asbestos fibers influences the protein secondary structure.Heliyon. 2024 Oct 4;10(20):e38966. doi: 10.1016/j.heliyon.2024.e38966. eCollection 2024 Oct 30. Heliyon. 2024. PMID: 39492902 Free PMC article.
-
The Effects of Asbestos Fibers on Human T Cells.Int J Mol Sci. 2020 Sep 23;21(19):6987. doi: 10.3390/ijms21196987. Int J Mol Sci. 2020. PMID: 32977478 Free PMC article. Review.
References
-
- Novello S., Pinto C., Torri V., Porcu L., Di Maio M., Tiseo M., Ceresoli G., Magnani C., Silvestri S., Veltri A., et al. The Third Italian Consensus Conference for Malignant Pleural Mesothelioma: State of the art and recommendations. Crit. Rev. Oncol. Hematol. 2016;104:9–20. doi: 10.1016/j.critrevonc.2016.05.004. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

