HIV-1 Vif's Capacity To Manipulate the Cell Cycle Is Species Specific
- PMID: 29321323
- PMCID: PMC5972884
- DOI: 10.1128/JVI.02102-17
HIV-1 Vif's Capacity To Manipulate the Cell Cycle Is Species Specific
Abstract
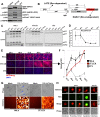
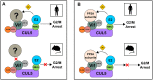
Cells derived from mice and other rodents exhibit profound blocks to HIV-1 virion production, reflecting species-specific incompatibilities between viral Tat and Rev proteins and essential host factors cyclin T1 (CCNT1) and exportin-1 (XPO1, also known as CRM1), respectively. To determine if mouse cell blocks other than CCNT1 and XPO1 affect HIV's postintegration stages, we studied HIV-1NL4-3 gene expression in mouse NIH 3T3 cells modified to constitutively express HIV-1-compatible versions of CCNT1 and XPO1 (3T3.CX cells). 3T3.CX cells supported both Rev-independent and Rev-dependent viral gene expression and produced relatively robust levels of virus particles, confirming that CCNT1 and XPO1 represent the predominant blocks to these stages. Unexpectedly, however, 3T3.CX cells were remarkably resistant to virus-induced cytopathic effects observed in human cell lines, which we mapped to the viral protein Vif and its apparent species-specific capacity to induce G2/M cell cycle arrest. Vif was able to mediate rapid degradation of human APOBEC3G and the PPP2R5D regulatory B56 subunit of the PP2A phosphatase holoenzyme in mouse cells, thus demonstrating that VifNL4-3's modulation of the cell cycle can be functionally uncoupled from some of its other defined roles in CUL5-dependent protein degradation. Vif was also unable to induce G2/M cell cycle arrest in other nonhuman cell types, including cells derived from nonhuman primates, leading us to propose that one or more human-specific cofactors underpin Vif's ability to modulate the cell cycle.IMPORTANCE Cells derived from mice and other rodents exhibit profound blocks to HIV-1 replication, thus hindering the development of a low-cost small-animal model for studying HIV/AIDS. Here, we engineered otherwise-nonpermissive mouse cells to express HIV-1-compatible versions of two species-specific host dependency factors, cyclin T1 (CCNT1) and exportin-1 (XPO1) (3T3.CX cells). We show that 3T3.CX cells rescue HIV-1 particle production but, unexpectedly, are completely resistant to virus-induced cytopathic effects. We mapped these effects to the viral accessory protein Vif, which induces a prolonged G2/M cell cycle arrest followed by apoptosis in human cells. Combined, our results indicate that one or more additional human-specific cofactors govern HIV-1's capacity to modulate the cell cycle, with potential relevance to viral pathogenesis in people and existing animal models.
Keywords: APOBEC3G; G2/M; HIV; PPP2R5D; Rev; Vif; cell cycle; cyclin T1; exportin-1; species specific.
Copyright © 2018 American Society for Microbiology.
Figures




Similar articles
-
Functional and Structural Insights into a Vif/PPP2R5 Complex Elucidated Using Patient HIV-1 Isolates and Computational Modeling.J Virol. 2020 Oct 14;94(21):e00631-20. doi: 10.1128/JVI.00631-20. Print 2020 Oct 14. J Virol. 2020. PMID: 32847850 Free PMC article.
-
Critical role of PP2A-B56 family protein degradation in HIV-1 Vif mediated G2 cell cycle arrest.Biochem Biophys Res Commun. 2020 Jun 18;527(1):257-263. doi: 10.1016/j.bbrc.2020.04.123. Epub 2020 May 4. Biochem Biophys Res Commun. 2020. PMID: 32446377
-
Vif-CBFβ interaction is essential for Vif-induced cell cycle arrest.Biochem Biophys Res Commun. 2019 Apr 16;511(4):910-915. doi: 10.1016/j.bbrc.2019.02.136. Epub 2019 Mar 7. Biochem Biophys Res Commun. 2019. PMID: 30851937
-
[Progress in the study of HIV-1 Vif and related inhibitors].Yao Xue Xue Bao. 2010 Jun;45(6):684-93. Yao Xue Xue Bao. 2010. PMID: 20939174 Review. Chinese.
-
Demystifying Cell Cycle Arrest by HIV-1 Vif.Trends Microbiol. 2021 May;29(5):381-384. doi: 10.1016/j.tim.2021.01.001. Epub 2021 Jan 19. Trends Microbiol. 2021. PMID: 33478820 Free PMC article. Review.
Cited by
-
HIV-1 Vif Triggers Cell Cycle Arrest by Degrading Cellular PPP2R5 Phospho-regulators.Cell Rep. 2019 Oct 29;29(5):1057-1065.e4. doi: 10.1016/j.celrep.2019.09.057. Cell Rep. 2019. PMID: 31665623 Free PMC article.
-
MiR-483-3p improves learning and memory abilities via XPO1 in Alzheimer's disease.Brain Behav. 2022 Aug;12(8):e2680. doi: 10.1002/brb3.2680. Epub 2022 Jul 14. Brain Behav. 2022. PMID: 35833267 Free PMC article.
-
Antagonism of PP2A is an independent and conserved function of HIV-1 Vif and causes cell cycle arrest.Elife. 2020 Apr 15;9:e53036. doi: 10.7554/eLife.53036. Elife. 2020. PMID: 32292164 Free PMC article.
-
Studying Retroviral Life Cycles Using Visible Viruses and Live Cell Imaging.Annu Rev Virol. 2024 Sep;11(1):125-146. doi: 10.1146/annurev-virology-100422-012608. Epub 2024 Aug 30. Annu Rev Virol. 2024. PMID: 38876144 Free PMC article. Review.
-
Primary infection with Zika virus provides one-way heterologous protection against Spondweni virus infection in rhesus macaques.Sci Adv. 2023 Jun 30;9(26):eadg3444. doi: 10.1126/sciadv.adg3444. Epub 2023 Jun 30. Sci Adv. 2023. PMID: 37390207 Free PMC article.
References
-
- Seay K, Qi X, Zheng JH, Zhang C, Chen K, Dutta M, Deneroff K, Ochsenbauer C, Kappes JC, Littman DR, Goldstein H. 2013. Mice transgenic for CD4-specific human CD4, CCR5 and cyclin T1 expression: a new model for investigating HIV-1 transmission and treatment efficacy. PLoS One 8:e63537. doi:10.1371/journal.pone.0063537. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

