Autophagy: A Potential Therapeutic Target for Reversing Sepsis-Induced Immunosuppression
- PMID: 29326712
- PMCID: PMC5741675
- DOI: 10.3389/fimmu.2017.01832
Autophagy: A Potential Therapeutic Target for Reversing Sepsis-Induced Immunosuppression
Abstract
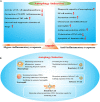
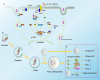
Sepsis remains the leading cause of mortality in intensive care units and an intractable condition due to uncontrolled inflammation together with immune suppression. Dysfunction of immune cells is considered as a major cause for poor outcome of septic patients but with little specific treatments. Currently, autophagy that is recognized as an important self-protective mechanism for cellular survival exhibits great potential for maintaining immune homeostasis and alleviating multiple organ failure, which further improves survival of septic animals. The protective effect of autophagy on immune cells covers both innate and adaptive immune responses and refers to various cellular receptors and intracellular signaling. Multiple drugs and measures are reportedly beneficial for septic challenge by inducing autophagy process. Therefore, autophagy might be an effective target for reversing immunosuppression compromised by sepsis.
Keywords: apoptosis; autophagy; immunosuppression; sepsis; treatment.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

