Overexpression of the double homeodomain protein DUX4c interferes with myofibrillogenesis and induces clustering of myonuclei
- PMID: 29329560
- PMCID: PMC5767009
- DOI: 10.1186/s13395-017-0148-4
Overexpression of the double homeodomain protein DUX4c interferes with myofibrillogenesis and induces clustering of myonuclei
Abstract
Background: Facioscapulohumeral muscular dystrophy (FSHD) is associated with DNA hypomethylation at the 4q35 D4Z4 repeat array. Both the causal gene DUX4 and its homolog DUX4c are induced. DUX4c is immunodetected in every myonucleus of proliferative cells, while DUX4 is present in only 1/1000 of myonuclei where it initiates a gene deregulation cascade. FSHD primary myoblasts differentiate into either atrophic or disorganized myotubes. DUX4 expression induces atrophic myotubes and associated FSHD markers. Although DUX4 silencing normalizes the FSHD atrophic myotube phenotype, this is not the case for the disorganized phenotype. DUX4c overexpression increases the proliferation rate of human TE671 rhabdomyosarcoma cells and inhibits their differentiation, suggesting a normal role during muscle differentiation.
Methods: By gain- and loss-of-function experiments in primary human muscle cells, we studied the DUX4c impact on proliferation, differentiation, myotube morphology, and FSHD markers.
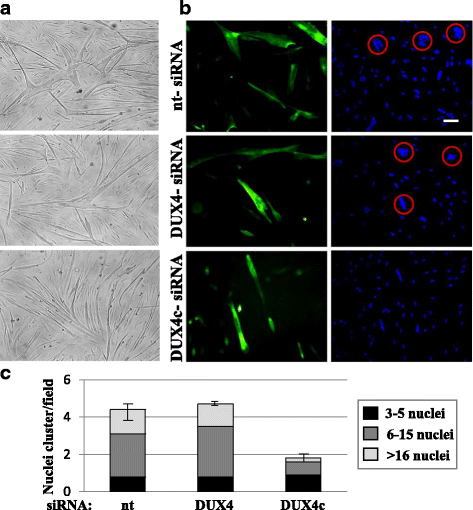
Results: In primary myoblasts, DUX4c overexpression increased the staining intensity of KI67 (a proliferation marker) in adjacent cells and delayed differentiation. In differentiating cells, DUX4c overexpression led to the expression of some FSHD markers including β-catenin and to the formation of disorganized myotubes presenting large clusters of nuclei and cytoskeletal defects. These were more severe when DUX4c was expressed before the cytoskeleton reorganized and myofibrils assembled. In addition, endogenous DUX4c was detected at a higher level in FSHD myotubes presenting abnormal clusters of nuclei and cytoskeletal disorganization. We found that the disorganized FSHD myotube phenotype could be rescued by silencing of DUX4c, not DUX4.
Conclusion: Excess DUX4c could disturb cytoskeletal organization and nuclear distribution in FSHD myotubes. We suggest that DUX4c up-regulation could contribute to DUX4 toxicity in the muscle fibers by favoring the clustering of myonuclei and therefore facilitating DUX4 diffusion among them. Defining DUX4c functions in the healthy skeletal muscle should help to design new targeted FSHD therapy by DUX4 or DUX4c inhibition without suppressing DUX4c normal function.
Keywords: Cytoskeleton; Differentiation; Disorganized myotubes; Myonuclear clustering; Proliferation; β-catenin.
Conflict of interest statement
Authors’ information
CV, AT, SC, and AW contributed to this manuscript during their Ph.D. or post-doctoral studies in the Laboratory of Molecular Biology. The current position of CV, AT, SC, and AW are respectively at SGS (Belgium), Laboratory of Respiratory Physiology and Rehabilitation (Research Institute for Health Sciences and Technology) at the University of Mons, GSK (Belgium) and Straticell (Belgium).
Ethics approval and consent to participate
Primary human myoblasts were derived from muscle biopsies performed according to the current ethical and legislative rules of France, and written informed consent was obtained from all subjects, as directed by the ethical committee of CHU de Villeneuve (Montpellier, France) (Barro et al. 2010). In addition, the use of this material was approved by the ethics committee of the University of Mons (ref # A901) and the ethics committee of ULB-Erasme (Brussels ref. #B2011/003 and #P2015/516).
Consent for publication
Prof. Peter S. Zammit gave us permission to refer to the data his group presented at the FSHD International Research Consortium 2009, Boston, Massachusetts.
Dr. Alexandre Philips (CRBM, CNRS, Montpellier), now retired from research, gave us permission to publish our collaborative study.
Competing interests
The authors (CV, EA, FC, and AB) are inventors in a patent application for antisense agents useful in treating FSHD and targeting either DUX4 or DUX4c (EP patent # 242 62 03, published in the European Patent Bulletin (March 7 2012) and US patent # US20120225034 A, published on September 12, 2012.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures





Similar articles
-
DUX4 expression in FSHD muscle cells: how could such a rare protein cause a myopathy?J Cell Mol Med. 2013 Jan;17(1):76-89. doi: 10.1111/j.1582-4934.2012.01647.x. Epub 2012 Dec 4. J Cell Mol Med. 2013. PMID: 23206257 Free PMC article.
-
The FSHD atrophic myotube phenotype is caused by DUX4 expression.PLoS One. 2011;6(10):e26820. doi: 10.1371/journal.pone.0026820. Epub 2011 Oct 28. PLoS One. 2011. PMID: 22053214 Free PMC article.
-
The double homeodomain protein DUX4c is associated with regenerating muscle fibers and RNA-binding proteins.Skelet Muscle. 2023 Mar 7;13(1):5. doi: 10.1186/s13395-022-00310-y. Skelet Muscle. 2023. PMID: 36882853 Free PMC article.
-
Deciphering transcription dysregulation in FSH muscular dystrophy.J Hum Genet. 2012 Aug;57(8):477-84. doi: 10.1038/jhg.2012.74. Epub 2012 Jun 21. J Hum Genet. 2012. PMID: 22718021 Free PMC article. Review.
-
Influence of DUX4 Expression in Facioscapulohumeral Muscular Dystrophy and Possible Treatments.Int J Mol Sci. 2023 May 30;24(11):9503. doi: 10.3390/ijms24119503. Int J Mol Sci. 2023. PMID: 37298453 Free PMC article. Review.
Cited by
-
Consequences of epigenetic derepression in facioscapulohumeral muscular dystrophy.Clin Genet. 2020 Jun;97(6):799-814. doi: 10.1111/cge.13726. Epub 2020 Mar 4. Clin Genet. 2020. PMID: 32086799 Free PMC article. Review.
-
High-resolution breakpoint junction mapping of proximally extended D4Z4 deletions in FSHD1 reveals evidence for a founder effect.Hum Mol Genet. 2022 Mar 3;31(5):748-760. doi: 10.1093/hmg/ddab250. Hum Mol Genet. 2022. PMID: 34559225 Free PMC article.
-
Identification of the hyaluronic acid pathway as a therapeutic target for facioscapulohumeral muscular dystrophy.Sci Adv. 2019 Dec 11;5(12):eaaw7099. doi: 10.1126/sciadv.aaw7099. eCollection 2019 Dec. Sci Adv. 2019. PMID: 31844661 Free PMC article.
-
Specialized Positioning of Myonuclei Near Cell-Cell Junctions.Front Physiol. 2018 Nov 1;9:1531. doi: 10.3389/fphys.2018.01531. eCollection 2018. Front Physiol. 2018. PMID: 30443220 Free PMC article.
-
Genome wide analysis of gene expression changes in skin from patients with type 2 diabetes.PLoS One. 2020 Feb 21;15(2):e0225267. doi: 10.1371/journal.pone.0225267. eCollection 2020. PLoS One. 2020. PMID: 32084158 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

