Engineering Polymersomes for Diagnostics and Therapy
- PMID: 29334183
- PMCID: PMC6377267
- DOI: 10.1002/adhm.201701276
Engineering Polymersomes for Diagnostics and Therapy
Abstract
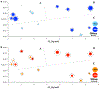
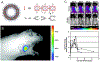
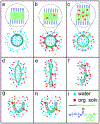
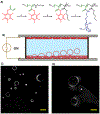
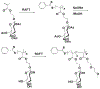
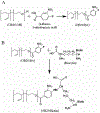
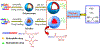
Engineered polymer vesicles, termed as polymersomes, confer a flexibility to control their structure, properties, and functionality. Self-assembly of amphiphilic copolymers leads to vesicles consisting of a hydrophobic bilayer membrane and hydrophilic core, each of which is loaded with a wide array of small and large molecules of interests. As such, polymersomes are increasingly being studied as carriers of imaging probes and therapeutic drugs. Effective delivery of polymersomes necessitates careful design of polymersomes. Therefore, this review article discusses the design strategies of polymersomes developed for enhanced transport and efficacy of imaging probes and therapeutic drugs. In particular, the article focuses on overviewing technologies to regulate the size, structure, shape, surface activity, and stimuli- responsiveness of polymersomes and discussing the extent to which these properties and structure of polymersomes influence the efficacy of cargo molecules. Taken together with future considerations, this article will serve to improve the controllability of polymersome functions and accelerate the use of polymersomes in biomedical applications.
Keywords: bilayer membrane; biomedical; drug delivery; self-assembly; vesicles.
© 2018 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures
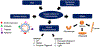









Similar articles
-
Nanoscale Polymersomes as Anti-Cancer Drug Carriers Applied for Pharmaceutical Delivery.Curr Pharm Des. 2016;22(19):2857-65. doi: 10.2174/1381612822666160217142319. Curr Pharm Des. 2016. PMID: 26898733 Free PMC article. Review.
-
Biocompatible polymersomes-based cancer theranostics: Towards multifunctional nanomedicine.Int J Pharm. 2017 Mar 15;519(1-2):287-303. doi: 10.1016/j.ijpharm.2017.01.037. Epub 2017 Jan 20. Int J Pharm. 2017. PMID: 28115259 Review.
-
Polymersomes: a new multi-functional tool for cancer diagnosis and therapy.Methods. 2008 Sep;46(1):25-32. doi: 10.1016/j.ymeth.2008.05.006. Epub 2008 Jun 20. Methods. 2008. PMID: 18572025 Free PMC article. Review.
-
Engineering Intracellular Delivery Nanocarriers and Nanoreactors from Oxidation-Responsive Polymersomes via Synchronized Bilayer Cross-Linking and Permeabilizing Inside Live Cells.J Am Chem Soc. 2016 Aug 24;138(33):10452-66. doi: 10.1021/jacs.6b04115. Epub 2016 Aug 10. J Am Chem Soc. 2016. PMID: 27485779
-
Stimuli-Responsive Polymersomes for Biomedical Applications.Biomacromolecules. 2017 Mar 13;18(3):649-673. doi: 10.1021/acs.biomac.6b01704. Epub 2017 Feb 17. Biomacromolecules. 2017. PMID: 28212005
Cited by
-
Membrane protein channels equipped with a cleavable linker for inducing catalysis inside nanocompartments.J Mater Chem B. 2021 Nov 10;9(43):9012-9022. doi: 10.1039/d1tb01463c. J Mater Chem B. 2021. PMID: 34623367 Free PMC article.
-
Polymeric Nanoparticles for Drug Delivery.Chem Rev. 2024 May 8;124(9):5505-5616. doi: 10.1021/acs.chemrev.3c00705. Epub 2024 Apr 16. Chem Rev. 2024. PMID: 38626459 Free PMC article. Review.
-
TME-activatable theranostic nanoplatform with ATP burning capability for tumor sensitization and synergistic therapy.Theranostics. 2020 May 25;10(15):6987-7001. doi: 10.7150/thno.44569. eCollection 2020. Theranostics. 2020. PMID: 32550917 Free PMC article.
-
Tumor Stimulus-Responsive Biodegradable Diblock Copolymer Conjugates as Efficient Anti-Cancer Nanomedicines.J Pers Med. 2022 Apr 27;12(5):698. doi: 10.3390/jpm12050698. J Pers Med. 2022. PMID: 35629120 Free PMC article.
-
Insights on Development Aspects of Polymeric Nanocarriers: The Translation from Bench to Clinic.Polymers (Basel). 2022 Aug 29;14(17):3545. doi: 10.3390/polym14173545. Polymers (Basel). 2022. PMID: 36080620 Free PMC article. Review.
References
-
- Wu NZ, Da D, Rudoll TL, Needham D, Whorton AR, Dewhirst MW, Cancer Res. 1993, 53, 3765. - PubMed
-
- Barenholz Y, J. Control. Release 2012, 160, 117. - PubMed
-
- Uziely B, Jeffers S, Isacson R, Kutsch K, Wei-Tsao D, Yehoshua Z, Libson E, Muggia FM, Gabizon A, J. Clin. Oncol 1995, 13, 1777. - PubMed
-
- Lee H, Larson RG, Biomacromolecules 2016, 17, 1757. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

