A Mitochondria-Specific Fluorescent Probe for Visualizing Endogenous Hydrogen Cyanide Fluctuations in Neurons
- PMID: 29337546
- PMCID: PMC6276372
- DOI: 10.1021/jacs.7b12545
A Mitochondria-Specific Fluorescent Probe for Visualizing Endogenous Hydrogen Cyanide Fluctuations in Neurons
Abstract
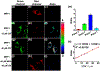
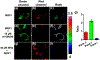
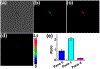
An ability to visualize HCN in mitochondria in real time may permit additional insights into the critical toxicological and physiological roles this classic toxin plays in living organisms. Herein, we report a mitochondria-specific coumarin pyrrolidinium-derived fluorescence probe (MRP1) that permits the real-time ratiometric imaging of HCN in living cells. The response is specific, sensitive (detection limit is ca. 65.6 nM), rapid (within 1 s), and reversible. Probe MRP1 contains a benzyl chloride subunit designed to enhance retention within the mitochondria under conditions where the mitochondria membrane potential is eliminated. It has proved effective in visualizing different concentrations of exogenous HCN in the mitochondria of HepG2 cells, as well as the imaging of endogenous HCN in the mitochondria of PC12 cells and within neurons. Fluctuations in HCN levels arising from the intracellular generation of HCN could be readily detected.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



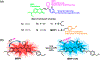
References
-
- Baskin SI; Brewer TG Cyanide poisoning In Medical Aspects of Chemical and Biological Warfare; Sidell F, Takafuji ET, Franz DR, Eds.; TMM Publication: Washington, DC, 1997; p 271.
-
- Mokhlesi B; Leikin JB; Murray P; Corbridge TC Chest 2003, 123, 897. - PubMed
-
- Young C; Tidwell L; Anderson C Cyanide: Social, Industrial, and Economic Aspects; Minerals, Metals, and Materials Society: Warrendale, 2001.
- Long L; Wang L; Wu Y; Gong A; Da Z; Zhang C; Han Z Chem. - Asian J 2014, 9, 3291. - PubMed
- Zdor RE J. Appl. Microbiol 2015, 118, 267. - PubMed
- Ferrari LA; Giannuzzi L Forensic Sci. Int 2015, 256, 46. - PubMed
- Surleva AR; Drochioiu G J. Chem. Educ 2013, 90, 1654.
- Moussa SG; Leithead A; Li S-M; Chan TW; Wentzell JJB; Stroud C; Zhang J; Lee P; Lu G; Brook JR; Hayden K; Narayan J; Liggio J Atmos. Environ 2016, 131, 185.
- Reade MC; Davies SR; Morley PT; Dennett J; Jacobs IC; Australian Resuscitation, C. Emerg. Med. Australas 2012, 24, 225. - PubMed
-
- Way JL Annu. Rev. Pharmacol. Toxicol 1984, 24, 451. - PubMed
-
- Yamamoto H.-a.; Mohanan PV Toxicology 2002, 179, 29. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

