Molecular Determinants of Response to Anti-Programmed Cell Death (PD)-1 and Anti-Programmed Death-Ligand 1 (PD-L1) Blockade in Patients With Non-Small-Cell Lung Cancer Profiled With Targeted Next-Generation Sequencing
- PMID: 29337640
- PMCID: PMC6075848
- DOI: 10.1200/JCO.2017.75.3384
Molecular Determinants of Response to Anti-Programmed Cell Death (PD)-1 and Anti-Programmed Death-Ligand 1 (PD-L1) Blockade in Patients With Non-Small-Cell Lung Cancer Profiled With Targeted Next-Generation Sequencing
Erratum in
-
Erratum.J Clin Oncol. 2018 Jun 1;36(16):1645. doi: 10.1200/JCO.2018.79.2796. J Clin Oncol. 2018. PMID: 31329702 Free PMC article.
Abstract
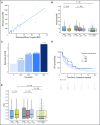
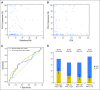
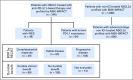
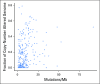
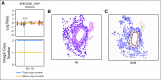
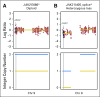
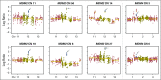
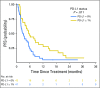
Purpose Treatment of advanced non-small-cell lung cancer with immune checkpoint inhibitors (ICIs) is characterized by durable responses and improved survival in a subset of patients. Clinically available tools to optimize use of ICIs and understand the molecular determinants of response are needed. Targeted next-generation sequencing (NGS) is increasingly routine, but its role in identifying predictors of response to ICIs is not known. Methods Detailed clinical annotation and response data were collected for patients with advanced non-small-cell lung cancer treated with anti-programmed death-1 or anti-programmed death-ligand 1 [anti-programmed cell death (PD)-1] therapy and profiled by targeted NGS (MSK-IMPACT; n = 240). Efficacy was assessed by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1, and durable clinical benefit (DCB) was defined as partial response/stable disease that lasted > 6 months. Tumor mutation burden (TMB), fraction of copy number-altered genome, and gene alterations were compared among patients with DCB and no durable benefit (NDB). Whole-exome sequencing (WES) was performed for 49 patients to compare quantification of TMB by targeted NGS versus WES. Results Estimates of TMB by targeted NGS correlated well with WES (ρ = 0.86; P < .001). TMB was greater in patients with DCB than with NDB ( P = .006). DCB was more common, and progression-free survival was longer in patients at increasing thresholds above versus below the 50th percentile of TMB (38.6% v 25.1%; P < .001; hazard ratio, 1.38; P = .024). The fraction of copy number-altered genome was highest in those with NDB. Variants in EGFR and STK11 associated with a lack of benefit. TMB and PD-L1 expression were independent variables, and a composite of TMB plus PD-L1 further enriched for benefit to ICIs. Conclusion Targeted NGS accurately estimates TMB and elevated TMB further improved likelihood of benefit to ICIs. TMB did not correlate with PD-L1 expression; both variables had similar predictive capacity. The incorporation of both TMB and PD-L1 expression into multivariable predictive models should result in greater predictive power.
Figures
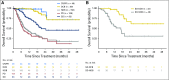
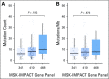

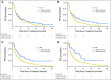
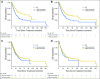


Comment in
-
Tumor Mutation Burden: Leading Immunotherapy to the Era of Precision Medicine?J Clin Oncol. 2018 Mar 1;36(7):631-632. doi: 10.1200/JCO.2017.76.8770. Epub 2018 Jan 16. J Clin Oncol. 2018. PMID: 29337637 No abstract available.
-
Targeted next-generation sequencing to assess tumor mutation burden: ready for prime-time in non-small cell lung cancer?Transl Lung Cancer Res. 2019 Dec;8(Suppl 4):S323-S326. doi: 10.21037/tlcr.2019.09.01. Transl Lung Cancer Res. 2019. PMID: 32038906 Free PMC article. No abstract available.
References
-
- Garon EB, Rizvi NA, Hui R, et al. : Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 372:2018-2028, 2015 - PubMed
-
- Fehrenbacher L, Spira A, Ballinger M, et al. : Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): A multicentre, open-label, phase 2 randomised controlled trial. Lancet 387:1837-1846, 2016 - PubMed
-
- Herbst RS, Baas P, Kim DW, et al. : Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): A randomised controlled trial. Lancet 387:1540-1550, 2016 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

