A novel orally available Syk/Src/Jak2 inhibitor, SKLB-850, showed potent anti-tumor activities in B cell lymphoma (BCL) models
- PMID: 29340070
- PMCID: PMC5762338
- DOI: 10.18632/oncotarget.22847
A novel orally available Syk/Src/Jak2 inhibitor, SKLB-850, showed potent anti-tumor activities in B cell lymphoma (BCL) models
Abstract
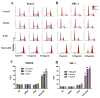
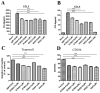
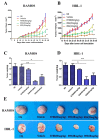
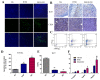
B cell lymphoma (BCL) is the most frequently diagnosed type of non-Hodgkin lymphoma (NHL), and accounts for about 4% of all cancers in the USA. Kinases spleen tyrosine kinase (Syk), Src, and Janus kinase 2 (JAK2) have been thought as potential targets for the treatment of BCL. We have recently developed a multikinase inhibitor, SKLB-850, which potently inhibits Syk, Src, and JAK2. The aim of this study is to investigate the anti-BCL activities and mechanisms of action of SKLB-850 both in vitro and in vivo. Our results showed that SKLB-850 significantly inhibited BCL cell proliferation, and induced apoptosis of BCL cells. It could considerably decrease the secretion of chemokines CCL3, CCL4, and CXCL12. Oral administration of SKLB-850 considerably suppressed the tumor growth in BCL xenograft models (Ramos and HBL-1) in a dose-dependent manner. Immunohistochemistry of tumor tissues showed that SKLB-850 efficiently inhibited the activation of Syk/ERK, Src/FAK and JAK2/Stat3 pathways. Collectively, SKLB-850 could be a promising agent for the treatment of BCL, hence deserving further study.
Keywords: B cell lymphoma; SKLB-850; multikinase inhibitor.
Conflict of interest statement
CONFLICTS OF INTEREST No potential conflicts of interest were disclosed.
Figures



References
-
- Muller AM, Ihorst G, Mertelsmann R, Engelhardt M. Epidemiology of non-Hodgkin’s lymphoma (NHL): trends, geographic distribution, and etiology. Ann Hematol. 2005;84:1–12. https://doi.org/10.1007/s00277-004-0939-7. - DOI - PubMed
-
- Tuccori M, Focosi D, Blandizzi C, Pelosini M, Montagnani S, Maggi F, Pistello M, Antonioli L, Fornai M, Pepe P, Rossi G, Petrini M. Inclusion of rituximab in treatment protocols for non-Hodgkin’s lymphomas and risk for progressive multifocal leukoencephalopathy. Oncologist. 2010;15:1214–9. https://doi.org/10.1634/theoncologist.2010-0098. - DOI - PMC - PubMed
-
- Alinari L, Quinion C, Blum KA. Bruton’s tyrosine kinase inhibitors in B-cell non-Hodgkin’s lymphomas. Clin Pharmacol Ther. 2015;97:469–77. https://doi.org/10.1002/cpt.65. - DOI - PubMed
-
- Ram R, Ben-Bassat I, Shpilberg O, Polliack A, Raanani P. The late adverse events of rituximab therapy--rare but there! Leuk Lymphoma. 2009;50:1083–95. https://doi.org/10.1080/10428190902934944. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

