AMPK Re-Activation Suppresses Hepatic Steatosis but its Downregulation Does Not Promote Fatty Liver Development
- PMID: 29343420
- PMCID: PMC5835560
- DOI: 10.1016/j.ebiom.2018.01.008
AMPK Re-Activation Suppresses Hepatic Steatosis but its Downregulation Does Not Promote Fatty Liver Development
Abstract
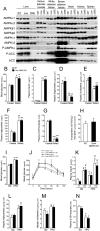
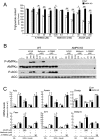
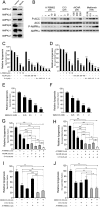
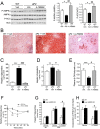
Nonalcoholic fatty liver disease is a highly prevalent component of disorders associated with disrupted energy homeostasis. Although dysregulation of the energy sensor AMP-activated protein kinase (AMPK) is viewed as a pathogenic factor in the development of fatty liver its role has not been directly demonstrated. Unexpectedly, we show here that liver-specific AMPK KO mice display normal hepatic lipid homeostasis and are not prone to fatty liver development, indicating that the decreases in AMPK activity associated with hepatic steatosis may be a consequence, rather than a cause, of changes in hepatic metabolism. In contrast, we found that pharmacological re-activation of downregulated AMPK in fatty liver is sufficient to normalize hepatic lipid content. Mechanistically, AMPK activation reduces hepatic triglyceride content both by inhibiting lipid synthesis and by stimulating fatty acid oxidation in an LKB1-dependent manner, through a transcription-independent mechanism. Furthermore, the effect of the antidiabetic drug metformin on lipogenesis inhibition and fatty acid oxidation stimulation was enhanced by combination treatment with small-molecule AMPK activators in primary hepatocytes from mice and humans. Overall, these results demonstrate that AMPK downregulation is not a triggering factor in fatty liver development but in contrast, establish the therapeutic impact of pharmacological AMPK re-activation in the treatment of fatty liver disease.
Keywords: AMPK; Fatty liver treatment; Lipid metabolism; Metformin; Nonalcoholic fatty liver disease (NAFLD); Small-molecule AMPK activators.
Copyright © 2018 The Authors. Published by Elsevier B.V. All rights reserved.
Figures




References
-
- Benziane B., Bjornholm M., Lantier L., Viollet B., Zierath J.R., Chibalin A.V. AMP-activated protein kinase activator A-769662 is an inhibitor of the Na(+)-K(+)-ATPase. Am. J. Physiol. Cell Physiol. 2009;297:C1554–1566. - PubMed
-
- Bergeron R., Previs S.F., Cline G.W., Perret P., Russell R.R., 3rd, Young L.H., Shulman G.I. Effect of 5-aminoimidazole-4-carboxamide-1-beta-D-ribofuranoside infusion on in vivo glucose and lipid metabolism in lean and obese Zucker rats. Diabetes. 2001;50:1076–1082. - PubMed
-
- Beynen A.C., Buechler K.F., van der Molen A.J., Geelen M.J. Inhibition of hepatic lipogenesis by salicylate. Toxicology. 1982;24:33–43. - PubMed
-
- Chalvon-Demersay T., Even P.C., Chaumontet C., Piedcoq J., Viollet B., Gaudichon C., Tome D., Foretz M., Azzout-Marniche D. Modifying the dietary carbohydrate-to-protein ratio alters the postprandial macronutrient oxidation pattern in liver of AMPK-deficient mice. J. Nutr. 2017;147:1669–1676. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

